 |
 |
- Search
| Obstet Gynecol Sci > Volume 59(5); 2016 > Article |
Abstract
Pregnant women with antiphospholipid syndrome (APS) carry a high risk of arterial or venous thrombosis. Such thrombotic conditions occur more frequently in patients with triple positivity to antiphospholipid antibodies or with high antibody titers. Hepatic infarction is a rare complication in pregnant women with APS, and it sometimes mimics HELLP syndrome. This report describes a preeclamptic pregnant woman with APS who had high titers of three antiphospholipid antibodies. She experienced severe epigastric pain with elevated liver enzymes; in addition, she had tachycardia and tachypnea. The clinical findings suggested hepatic infarction and pulmonary thromboembolism, a partial manifestation of catastrophic APS. Therefore, she underwent emergent cesarean section at 25+2 weeks of gestation. After the delivery, her laboratory test indicated HELLP-like features, and computed tomography confirmed hepatic infarction and pulmonary micro-thromboembolism. Here, we report a case of a partial manifestation of catastrophic APS in a pregnant woman with triple antibody positivity, including a brief literature review.
Antiphospholipid syndrome (APS) is an autoimmune disease that is characterized by venous or arterial thrombosis, or obstetric manifestations with antiphospholipid antibodies (aPL) [1,2]. Obstetric manifestations include recurrent pregnancy loss and preterm delivery that are complicated with early-onset preeclampsia or fetal growth restriction (FGR) related with uteroplacental insufficiency [1,2,3]. Positive results of aPL tests, including anticardiolipin antibody, ╬▓2-glycoprotein antibody, and lupus anticoagulant, are necessary to diagnose APS at 12 weeks interval [1,2]. If patient was showed positive result to all these antibodies, it called triple positivity. In addition, triple positivity to or high titers of aPL increase the risk of thromboembolism and adverse pregnancy outcomes [1,2,3,4].
Catastrophic APS was first defined in 1992 as a life-threatening variant of APS. It is characterized by multiorgan failure, caused by multiple small-vessel thromboses, occurring in a brief time [5,6]. Pregnancy is one of the risk factors of catastrophic APS, and it occurs more frequently with triple antibody positivity or with high antibody titers [2,7,8].
Here, we report on a pregnant woman with APS who had triple antibody positivity with high titers, and was complicated with a partial manifestation of catastrophic APS.
This case report describes a 35-year-old woman, gravida 1, para 0, who was diagnosed as APS. At her first pregnancy, she was referred to our hospital because of severe FGR, oligohydramnios, and chronic hypertension at 17 weeks of gestation. Multiple serum markers were elevated in the quad test (alpha-fetoprotein, 8.273 multiples of the median [MoM]; human chorionic gonadotropin, 1.396 MoM; inhibin-A, 7.321 MoM), implicating possible uteroplacental insufficiency. On the basis of her clinical features, APS was suspected and subsequent laboratory tests confirmed the diagnosis. Treatment with low-dose aspirin (LDA, 100 mg daily) and low molecular weight heparin (LMWH, enoxaparin 40 mg daily) was started. Nonetheless, her first pregnancy ended at 21 weeks of gestation because of fetal death in utero.
During her second pregnancy, treatment with high-dose LMWH (enoxaparin 40 mg, twice a day) and LDA was started from 6 weeks of gestation, considering her previous pregnancy loss and triple antibody positivity with high titers (Table 1). At 16 weeks, elevation of multiple serum makers (alpha-fetoprotein, 2.93 MoM; human chorionic gonadotropin, 5.44 MoM; inhibin A, 6.75 MoM) was found again. Furthermore, lagging of fetal growth was observed (283 g, 19 weeks sized) at 20 weeks. Considering her previous pregnancy loss history, triple antibody positivity with high titers, and delayed fetal growth despite anticoagulation, we decided to start treatment with intravenous heparin target activated partial thromboplastin time to improve uteroplacental microcirculation.
At 24+2 weeks, she experienced sudden-onset epigastric pain with aggravated hypertension (153/99 mmHg) despite on-going antihypertensive medication and newly developed proteinuria (24-hours urine protein 671.0 mg), indicating superimposed preeclampsia. Her aspartate aminotransferase and alanine aminotransferase (AST/ALT) levels were slightly elevated to 69/65 IU/L, which worsened to 106/199 IU/L the next day (Fig. 1). Taking these result together, we suspected severe preeclampsia or heparin-induced hepatotoxicity. Intramuscular betamethasone was administered for fetal lung maturation in case of imminent delivery. Administration of intravenous heparin continued but the target activated partial thromboplastin time level was lowered to 80 seconds. After 3 days, her epigastric pain had subsided and AST/ALT levels were normalized to 25/53 IU/L, simultaneously.
However, at 25+1 weeks, she again developed severe acute epigastric pain with fever, and her AST/ALT levels started to increase again (Fig. 1). On the basis of her symptoms, she underwent magnetic resonance cholangiography (MRI), considering cholecystitis and hepatic infarction. On MRI, hepatic infarction is usually appeared as peripheral wedge shaped lesion on T1 and T2 weighted images. However, MRI showed several oval shaped hyporesonance lesions, indicating tumors or abscess in segments 2 (S2) and 5 (S5) (Fig. 2). On the basis of these radiologic findings, empirical antibiotic therapy was started because of a suspected diagnosis of liver abscess. Despite the antibiotic treatment, her epigastric pain did not subside at all and indicated an ischemic nature; furthermore, level of AST/ALT was elevated to 236/305 IU/L and lactate dehydrogenase increased to 862 IU/L (Fig. 1). Moreover, acute onset of tachypnea (respiration rate 30-40/min), tachycardia (heart rate >120 bpm), and lowered O2 saturation occurred concurrently, and her condition rapidly deteriorated. Accordingly, her clinical findings suggested pulmonary thromboembolism and hepatic infarction, as a partial manifestation of catastrophic APS, as well as severe preeclampsia with HELLP syndrome.
Because of her severe condition as stated above, emergent cesarean section was performed and a neonate with a birth weight of 530 g was delivered with an Apgar score of 4 at 1 minute and 5 at 5 minutes. Eighteen hours after the surgery, we re-administered intravenous heparin. At postoperative 1 day, her epigastric pain had subsided considerably; however, her AST/ALT levels increased to 531/910 IU/L. In addition, lactate dehydrogenase was elevated to 1189 IU/L and platelet count decreased to 53,000 ┬ĄL (Fig. 1), indicating HELLP syndrome. At postoperative day 3, computed tomography (CT) of the liver and lung proved hepatic infarction in most parts of the liver S2 and S5, and pulmonary microthrombosis (Fig. 2). Eleven days after the operation, her epigastric pain was completely alleviated and she was discharged with anticoagulant. Her placental pathology revealed increased syncytial knots and intervillous fibrin, indicating uteroplacental hypoperfusion; however, definite vascular thrombosis was not observed. A liver CT at 3 weeks after discharge demonstrated that the extent of infarction had decreased (Fig. 2), and her baby showed no evidence of developmental delay at the 9-month follow-up.
Physiologically, pregnant women have a hypercoagulable tendency owing to venous stasis and alteration of thrombotic factors and the fibrinolytic system. These unique characteristics of pregnancy aggravate the risk of thromboembolism in pregnant women with APS [2,7,9]. Usually, thromboembolisms in APS patients commonly occur in the cerebrovascular or deep venous system with a 20% prevalence rate [2]. In contrast, abdominopelvic manifestations of thromboembolisms in APS patients are rarely reported and mostly present as Budd-Chiari syndrome [9]. Since the first report by Mor et al. in 1989 [10], hepatic infarction in pregnant women with APS was seldom reported because the dual blood supply system of the liver (portal vein and hepatic artery) protects against ischemic threat [9,10].
Preeclampsia (11% to 40% vs. 5% to 7%) and HELLP syndrome (10.8% vs. 0.01% to 0.8%) occur more often in APS patients than in the general population [2,4,9,11]. Interestingly, most of the reported cases of hepatic infarction in pregnant women with APS (15 of 16, 93%) were associated with HELLP syndrome [11]. In addition, these complications appeared earlier and with more severe features with APS [2,4,12].
Like our case, APS patients with triple positivity or high titers of antibodies are at a higher risk for poor pregnancy outcomes and vascular thrombosis than those without these conditions [1,2,4,12]. According to the European Registry on Obstetric Antiphospholipid Syndrome, pregnant women with triple positivity had an increased risk for miscarriages (odds ratio [OR], 1.22; 95% confidence interval [CI], 1.049 to 3.382), fetal loss (OR, 6.880; 95% CI, 1.9 to 24.914), and FGR (OR, 1.600; 95% CI, 1.017 to 1.251). In particular, patients with lupus anticoagulant carry higher risks of early-onset preeclampsia and HELLP (OR, 2.882; 95% CI, 1.496 to 5.326) and FGR (OR, 2.538; 95% CI, 1.197 to 5.382) than those with other aPL combinations [4].
Frequent prenatal checkup, every 2 to 4 weeks before midgestation and every 1 to 2 weeks thereafter, was proposed to reduce the adverse pregnancy outcomes [2]. No optimal treatment strategy for pregnant women with APS has been established. However, LMWH and LDA are usually recommended as the treatment options in pregnant women with APS for prophylaxis or therapeutic purposes [2,3,4]. In general, a regimen with LMWH (40 mg/day) and LDA (100 mg/day) is frequently used. However, it is recommended to start LMWH administration with high dosage (1 mg/kg twice a day or 1.5 mg/kg daily) during the first trimester in pregnant women with a previous thrombosis or a history of recurrent pregnancy loss [2,3,4,12].
Besides the conventional therapy stated above, other remedies have also been attempted, including glucocorticoid, intravenous immunoglobulins (IVIG), and plasma exchange therapy, which are prescribed to control antibody activities [2,4]. However, corticosteroids and IVIG were found to have no benefit compared with anticoagulant. Moreover, corticosteroid shows adverse effects such as maternal diabetes, hypertension, and prematurity with long-term use. Therefore, corticosteroids and IVIG are used additively or in patients who were refractory to anticoagulation [2,3,5]. Intriguingly, De Carolis et al. [13] recently reported that aPL titers in pregnant women could be reduced with hydroxychloroquine medication. Moreover, a significant decrease of pregnancy loss, from 81% to 19%, was demonstrated after the addition of hydroxychloroquine therapy to refractory obstetric APS patients [14]. Based on these observations, adding hydroxychloroquine to anticoagulant was proposed for high-risk patients with triple positivity with high titers or refractory obstetric APS [14]. Nonetheless, when fatal maternal or fetal complication still occurs despite all these treatments, termination of pregnancy is mandatory to minimize maternal and neonatal complication [2,3,5,7].
Catastrophic APS, also known as Asherson's syndrome, is a lethal and extremely rare variant of APS (mortality rate 46% to 50%) and diagnosed when thrombosis had occurred in at least three organs within 1 week [5,6,7,8]. Catastrophic APS is triggered by infection, trauma, surgery, and pregnancy [5,6]. Only 15 cases of catastrophic APS have been reported in pregnant women after a registry for catastrophic APS was created by the European Forum on Antiphospholipid Antibodies [7]. Gomez-Puerta et al. [7] analyzed these patients and found that catastrophic APS showed specific features similar to HELLP syndrome in half of the cases, among them, hepatic infarction occurred in three cases. Therefore, it seems important to consider the possibility of catastrophic APS and hepatic infarction in pregnant women with signs of HELLP syndrome [7,8].
When catastrophic APS is suspected, imaging studies are recommended to detect thrombosis [5,6,7,8]. Representatively, if hepatic infarction is suspected, MRI and CT are useful modality for diagnosis. They are show peripheral wedge shape lesion with high signal on T2-weighted images and low signal on T1-weighted images through MRI and with low attenuation through CT scan [15].
For the treatment of catastrophic APS, three goals are considered: treating the precipitating factor, starting anticoagulation, and preventing cytokine storm [2,5,6,7,8]. However, the cornerstone for the treatment of catastrophic APS is not yet determined. Therefore, if catastro-phic APS is suspected, anticoagulation and empirical treatments, as discussed above, should be conducted [2,5,6,7,8].
Our case report is meaningful because it is first reported case of partial catastrophic APS in pregnant woman in Korea. Unlike other cases, our patient was diagnosed as partial catastrophic APS with hepatic infarction and pulmonary microthromboembolism which were presented rarely. Despite early prophylaxis, hepatic infarction and pulmonary micro-thromboembolism occurred. However, it did not mean to, betamethasone administration for fetal lung maturartion improved patient symptom and laboratory result for a while. Although, MRI was not helpful for diagnosis, her clinical signs and symptoms indicated hepatic infarction and postnatal CT confirmed the diagnosis.
In conclusion, hepatic infarction and catastrophic APS in pregnant women are extremely rare and difficult to diagnose because of substantial overlapping with other pregnancy complications such as preeclampsia or HELLP syndrome. Therefore, this report provides useful information on hepatic infarction and catastrophic APS in pregnant women.
References
1. Bertolaccini ML, Khamashta MA. Laboratory diagnosis and management challenges in the antiphospholipid syndrome. Lupus 2006;15:172-178. PMID: 16634372.


2. Ruiz-Irastorza G, Crowther M, Branch W, Khamashta MA. Antiphospholipid syndrome. Lancet 2010;376:1498-1509. PMID: 20822807.


3. Erkan D, Aguiar CL, Andrade D, Cohen H, Cuadrado MJ, Danowski A, et al. 14th International Congress on Antiphospholipid Antibodies: task force report on antiphospholipid syndrome treatment trends. Autoimmun Rev 2014;13:685-696. PMID: 24468415.


4. Alijotas-Reig J, Ferrer-Oliveras R, Ruffatti A, Tincani A, Lefkou E, Bertero MT, et al. The European Registry on Obstetric Antiphospholipid Syndrome (EUROAPS): a survey of 247 consecutive cases. Autoimmun Rev 2015;14:387-395. PMID: 25555817.


5. Asherson RA, Cervera R, de Groot PG, Erkan D, Boffa MC, Piette JC, et al. Catastrophic antiphospholipid syndrome: international consensus statement on classification criteria and treatment guidelines. Lupus 2003;12:530-534. PMID: 12892393.


6. Asherson RA, Cervera R, Piette JC, Shoenfeld Y, Espinosa G, Petri MA, et al. Catastrophic antiphospholipid syndrome: clues to the pathogenesis from a series of 80 patients. Medicine (Baltimore) 2001;80:355-377. PMID: 11704713.


7. Gomez-Puerta JA, Cervera R, Espinosa G, Asherson RA, Garcia-Carrasco M, da Costa IP, et al. Catastrophic antiphospholipid syndrome during pregnancy and puerperium: maternal and fetal characteristics of 15 cases. Ann Rheum Dis 2007;66:740-746. PMID: 17223653.



8. Hanouna G, Morel N, Le Thi Huong D, Josselin L, Vauthier-Brouzes D, Saadoun D, et al. Catastrophic antiphospholipid syndrome and pregnancy: an experience of 13 cases. Rheumatology (Oxford) 2013;52:1635-1641. PMID: 23676524.


9. Pauzner R, Dulitzky M, Carp H, Mayan H, Kenett R, Farfel Z, et al. Hepatic infarctions during pregnancy are associated with the antiphospholipid syndrome and in addition with complete or incomplete HELLP syndrome. J Thromb Haemost 2003;1:1758-1763. PMID: 12911590.


10. Mor F, Beigel Y, Inbal A, Goren M, Wysenbeek AJ. Hepatic infarction in a patient with the lupus anticoagulant. Arthritis Rheum 1989;32:491-495. PMID: 2495803.


11. Le Thi Thuong D, Tieulie N, Costedoat N, Andreu MR, Wechsler B, Vauthier-Brouzes D, et al. The HELLP syndrome in the antiphospholipid syndrome: retrospective study of 16 cases in 15 women. Ann Rheum Dis 2005;64:273-278. PMID: 15647435.



12. Hernandez-Molina G, Espericueta-Arriola G, Cabral AR. The role of lupus anticoagulant and triple marker positivity as risk factors for rethrombosis in patients with primary antiphospholipid syndrome. Clin Exp Rheumatol 2013;31:382-388. PMID: 23305633.

13. De Carolis S, Botta A, Salvi S, di Pasquo E, Del Sordo G, Garufi C, et al. Is there any role for the hydroxychloroquine (HCQ) in refractory obstetrical antiphospholipid syndrome (APS) treatment? Autoimmun Rev 2015;14:760-762. PMID: 25936295.


14. Mekinian A, Lazzaroni MG, Kuzenko A, Alijotas-Reig J, Ruffatti A, Levy P, et al. The efficacy of hydroxychloroquine for obstetrical outcome in anti-phospholipid syndrome: data from a European multicenter retrospective study. Autoimmun Rev 2015;14:498-502. PMID: 25617818.


15. Guiu B, Loffroy R, Cercueil JP, Sagot P, Krause D, Tixier H. MRI diagnosis and follow-up of hepatic infarction in a patient with antiphospholipid syndrome in early pregnancy. Arch Gynecol Obstet 2011;283:659-662. PMID: 20411270.


Fig.┬Ā1
Laboratory result of our patient at her second pregnancy. AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; ACS, antenatal corticosteroid; GA, gestational age; POD, post-operative day.
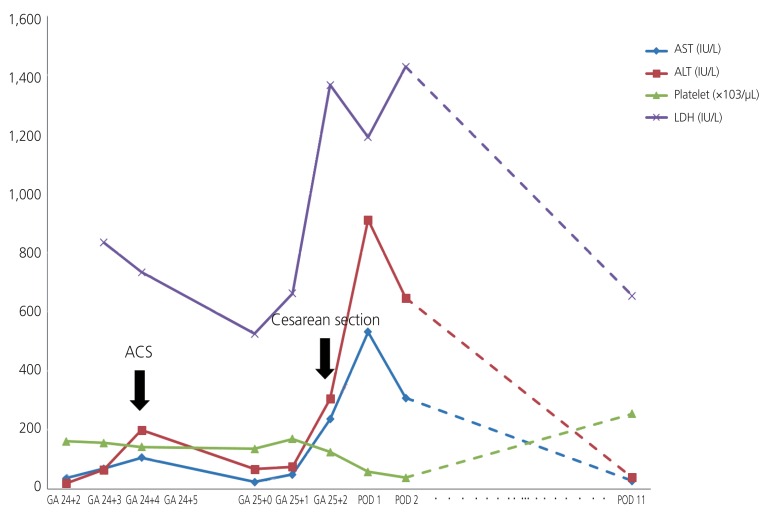
Fig.┬Ā2
Radiologic finding of our patient at her second pregnancy. (A) Magnetic resonance cholangiography; at 25+1 weeks of gestation, the day before delivery, 4.5-cm-sized clustered appearance in liver segment 2 which indicated to liver abscess (arrowed). (B) Pulmonary embolism computed tomography; postoperative 1 day, revered halo sign was seen in both lower lung field which indicated pulmonary micro-thromboembolism (arrow head). (C-F) Contrast enhanced liver computed tomography, axial and coronal view. (C,D) Postoperative 4 day, non-enhanced parenchymal peripheral wedge shaped lesion involving liver segment 2 and segment 5, which indicated hepatic infarction (arrowed). (E,F) After 4 month from cesarean section, hypotrophy of liver segment 2 and decreased size of infarcted area of liver showed.

Table┬Ā1
Antiphospholipid antibodies and other serological data of our patient at her second pregnancy
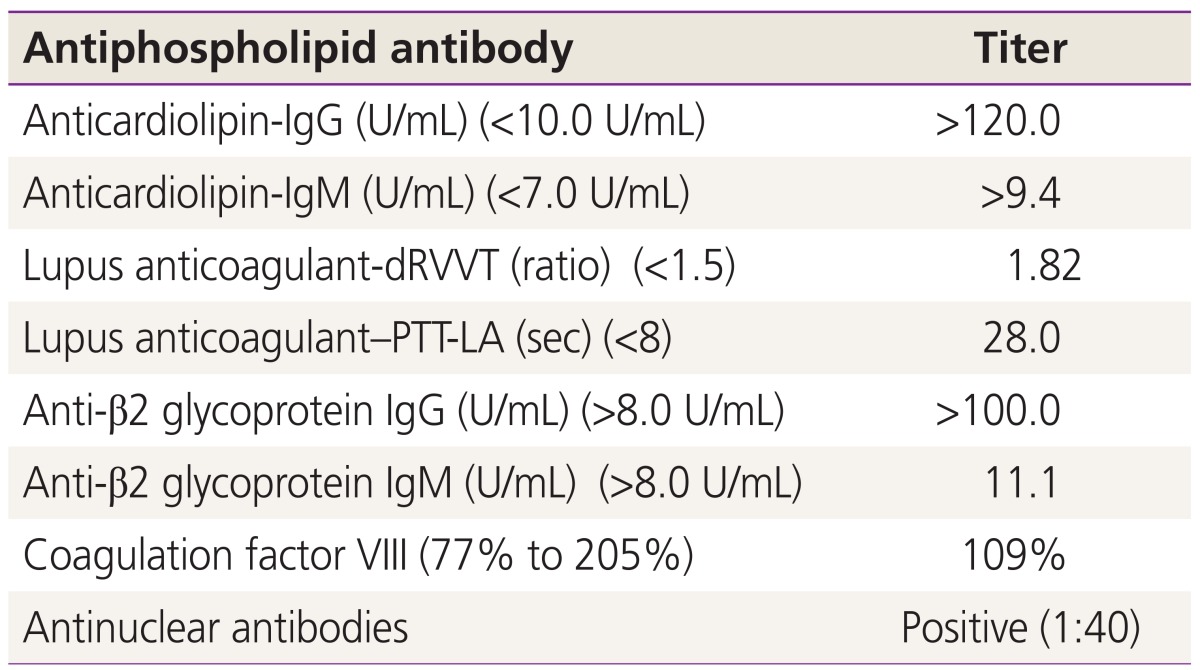
This table shows triple positivity to all antiphospholipid antibodies; All three antibodies showed a high titer; Values in parentheses are normal values.
IgG, immunoglobulin G; IgM, immunoglobulin M; dRVVT, dilute Russell viper venom time; PTT-LA, partial thromboplastin time reagents sensitive for the detection of lupus anticoagulant.
-
METRICS

-
- 3 Crossref
- 6,284 View
- 34 Download
- Related articles in Obstet Gynecol Sci




















