|
 |
- Search
| Obstet Gynecol Sci > Volume 59(6); 2016 > Article |
Abstract
Invasive mole is a benign gestational trophoblastic disease that arises from the myometrial invasion of any gestational event via direct extension through tissue or vascular structures. Invasive mole (and other gestational trophoblastic diseases) may present with life-threatening complications including uterine perforation, excessive bleeding, acute hemoperitoneum, and abdominal pain. We report a case of invasive mole presenting as abdominal distention in a 51-year-old perimenopausal woman (gravida 12, para 12, abortion 0). The patient was admitted to the gynecology clinic with a giant uterine mass filling the pelvic and abdominal cavity. To our knowledge, this is the first case in the literature of a gestational trophoblastic neoplasia presenting with uterine mass of 28 weeks' gestational size in this age group. Interestingly, complications such as uterine rupture or invasion of the adjacent structures (such as parametrial tissues or blood vessels) had not developed in our patient despite the considerable enlargement of the uterus.
Invasive mole, a rare subgroup of gestational trophoblastic disease (GTD), is a highly curable and usually locally invasive form of malignancy. Invasive mole usually occurs in women of reproductive age, and is extremely rare in perimenopausal women [1].
Invasive moles are usually diagnosed clinically rather than pathologically when the human chorionic gonadotropin (hCG) levels plateau or rise during follow-up after molar evacuation. Invasive mole often arises after molar pregnancies, but can also rarely occur after any gestation, including miscarriages and term pregnancies. The most common presenting symptoms of the invasive mole are abnormal uterine bleeding and an enlarged uterus [2]. However, abnormal uterine bleeding may be masked by irregular menstrual cycles, which are commonly seen during the perimenopausal years. In addition, GTDs are usually considered a disease of the reproductive period, potentially leading to delays in diagnosis in perimenopausal women. Therefore, the disease may present with severe clinical manifestations such as uterine rupture, acute hemoperitoneum, and abdominal pain [3].
We report a case of invasive mole presenting as abdominal distention in a perimenopausal woman. Interestingly, despite enlargement of the uterus of up to 8 cm above the umbilicus (approximate size at 28 weeks' gestation), complications such as uterine rupture and invasion of the adjacent organs or vascular structures had not developed in our case.
A 51-year-old perimenopausal woman (gravida 12, para 12, abortion 0) was admitted to the gynecology clinic with abdominal distention. Her last pregnancy had been 12 years ago and had resulted in delivery at term. During the past 2 to 3 years she had suffered from oligomenorrhoea, with her last menstruation occurring 10 months before admission to the clinic. She had a 2-year history of spotting episodes (light, irregular bleeding). Contrary to the usual symptomatology, she had not had any other clinical symptom beyond abdominal distention and vaginal spotting. The patient had no history of contraceptive use or of GTD. Her blood type was O, Rh positive.
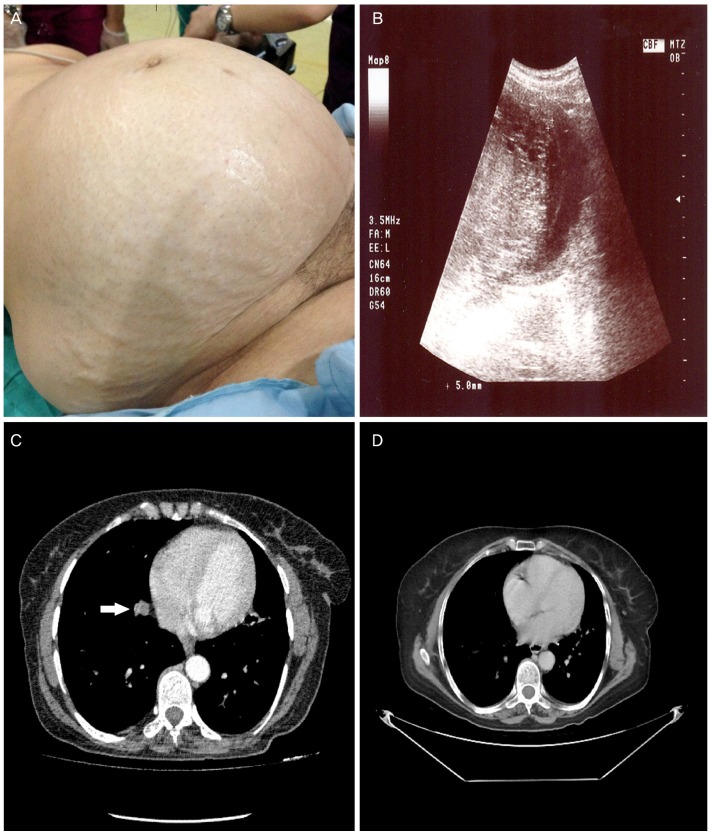
On gynecologic examination, a solid mass extending up to 8 cm above the umbilicus was palpated (Fig. 1A). The ultrasonographic scan confirmed that the giant mass originates from the uterus (Fig. 1B). The uterus was enlarged with a heterogeneous vesicular mass, obliterating the endometrial cavity to the approximate size of a 28-week gestation. Serum hCG was determined to be greater than 200,000 mIU/mL. Hepatic, renal, and thyroid function tests showed normal results. No abnormalities other than an enlarged uterus were observed on cranial or abdominal magnetic resonance images. Computerized tomographic images of the thorax showed multiple nodular lesions in both basal lung segments (Fig. 1C, D). An endometrial biopsy was considered, but because of her age and risk of sudden-onset uncontrollable vaginal hemorrhage, hysterectomy was considered a safer option.
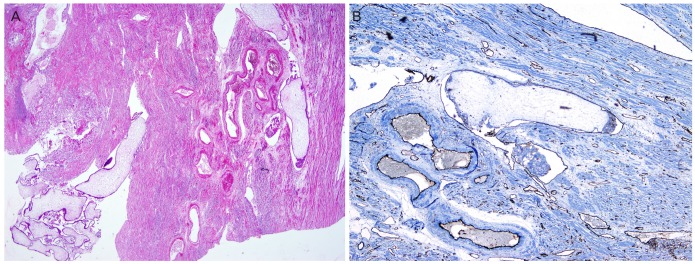
Exploratory laparotomy showed an evident thinning of the uterine wall and marked vascularity extending to the serosa of the uterus. Total abdominal hysterectomy and bilateral salpingo-oophorectomy were performed. During surgery, it was noted that the vessels in the parametrial region demonstrated increased fragility. The estimated blood loss was about 700 to 800 mL; an intraoperative transfusion was not necessary. Macroscopic findings confirmed an enlarged uterus (28×25×15 cm in size) and an enlarged endometrial cavity containing edematous villi. In our case, gross appearance strongly suggested a complete mole, but histopathologic and immunohistochemical examinations confirmed the diagnosis of the invasive mole. Microscopic examination revealed generalized hydropic villi and excessive proliferation of atypical and hyperchromatic trophoblastic cells, which invade into the adjacent myometrium or lymphatic vessels (Fig. 2A). Immunohistochemical studies showed that myometrial lymphatic vessels stained strongly with CD31 (Fig. 2B).
According to the World Health Organization modified prognostic scoring system for GTD, the patient was classified as high risk (total score 7); the patient was stage III on the International Federation of Gynecology and Obstetrics (FIGO) staging system. The patient's serum hCG levels dropped rapidly to 7,165 mIU/mL at the fifth week after surgery. Thereafter, 6 weeks after the operation, hCG levels declined to 2,962 mIU/mL and remained at a plateau between 2,500 to 3,500 mIU/ml during weekly follow-up evaluations (for a non-pregnant woman, the normal value is <5 mIU/mL). Combination chemotherapy regimen including EMA-CO (etoposide, methotrexate, and dactinomycin with cyclophosphamide and vincristine) was scheduled. Her hCG levels decreased to normal reference ranges in the second chemotherapy cycle; two additional cycles were given in order to reduce the risk of relapse. Her invasive mole went into complete remission as of follow-up 12 months after chemotherapy.
Hydatidiform moles are benign diseases with malignant potential. Malignant transformation occurs in 15% to 20% of complete hydatidiform moles and 1% to 5% of partial hydatidiform moles [4]. Invasive mole is a form of gestational trophoblastic neoplasia (GTN) that most commonly occurs after the evacuation of a molar pregnancy and is characterized by the presence of edematous chorionic villi with trophoblastic proliferation that invades into the myometrium or to adjacent structures such as the parametrium, vagina, and broad ligament. Invasive mole can also penetrate the uterine wall, causing uterine rupture and hemoperitoneum [3,5]. Most cases are clinically rather than histologically diagnosed. The clinical presentation of an invasive mole includes abnormal vaginal bleeding, high serum hCG levels, an enlarged uterus, and the existence of these symptoms after the evacuation of a molar pregnancy. Initial presentation as an invasive mole is extremely rare; it is preceded by hydatidiform mole in about 95% of cases. The interval from an antecedent molar pregnancy to the diagnosis of the invasive mole is usually less than 6 months [6].
Invasive mole generally occurs in women of reproductive age and is extremely rare in perimenopausal women. Here, we present the unusual and interesting clinical course of a patient with perimenopausal invasive mole. First, there had been no history of GTD during her reproductive years, whereas invasive mole usually occurs after molar pregnancy. Second, the invasive mole surprisingly did not lead to a severe complication such as uterine perforation, hemoperitoneum, or massive bleeding, in spite of the giant size of the uterus and insidious clinical course. The cause of the delayed diagnosis in our case was due to the absence of obvious clinical symptoms except for spotting episodes during the previous 2 years. However, the patient had experienced light, skipped, or late menstrual periods that had not been reported previously to a healthcare provider. Thus, the disease continued to progress until the patient was admitted to the gynecology clinic with abdominal distention. The uterus was enlarged to approximately 28 weeks' gestational size and was measured as 28×25×15 cm on ultrasonography. Her high parity may have allowed for expansion of the uterus up to such a large size. To our knowledge, this is the first case in the literature of a GTN reaching this uterine size in this age group.
The pathogenesis of GTNs in perimenopausal women is not exactly known, but it is known that ovulation may rarely occur in the perimenopausal and even postmenopausal women. Spontaneous ovulation in the perimenopausal woman resulting in the production of immature ova followed by defective fertilization may be the initial pathology in the development of GTNs [7]. In our case, the gross appearance of uterine content suggests a diagnosis of complete mole; the disease initially may have begun as a complete mole and then may have progress to the invasive mole.
The most common presenting symptoms of the invasive mole are irregular vaginal bleeding and an enlarging uterus. However, it can also cause serious complications during the enlargement of the uterus such as uterine perforation with massive hemoperitoneum, abdominal pain, uncontrollable hemorrhage, shock, and the need for emergency laparotomy. Untreated invasive moles tend to invade the uterine wall locally, which can result in uterine perforation and significant hemorrhage. These clinical presentations of invasive mole have been reported in patients of reproductive age [8,9].
There have been very few case reports of invasive mole in postmenopausal women. Invasive mole cases reported in the literature are usually treated by extirpative procedures or hysterectomy and then followed up with serial hCG levels [10]. Hysterectomy has been performed as an initial treatment in women during menopause. Taskin et al. [7] described a 53-year-old postmenopausal woman who presented with abdominal pain due to an enlarged uterus up to the size of 16 weeks' gestation. In their case, there was no evidence of metastasis beyond the uterus, and the patient was successfully treated with hysterectomy and bilateral salpingo-oophorectomy without additional therapy. Lok et al. [11] reported a case of a 56-year-old woman who developed hydatidiform mole. In this case, the uterus had enlarged to 24-weeks' gestation size, but no metastatic disease had been found in radiological scanning. They performed hysterectomy, and remission was maintained over 2 years of follow-up. In a review of 10 studies involving a total of 203 cases of GTD in older women, von Welser et al. [12] reported that hysterectomy should be considered as an option with a number of advantages in different situations. These researchers also pointed out that hysterectomy might be associated with increased blood loss due to increased vascularity. de la Fouchardiere et al. [13] reported a postmenopausal invasive mole following a pregnancy that occurred after a biologically confirmed menopause. In their patient, the pregnancy was confirmed by an ovarian corpus luteum. These authors explained the pathogenesis of postmenopausal invasive mole by a possible current pregnancy or malignant changes of trophoblastic remnants of a prior pregnancy after a period of latency. Tsukamoto et al. [14] reported 20 cases of GTD in women aged 50 years or more. The pathologies of these patients were 8 invasive moles (40%), 7 hydatidiform moles (35%), and 5 choriocarcinomas (25%). However, none of the patients with invasive mole was postmenopausal. The most common presenting symptom of the cases was abnormal vaginal bleeding. Because of the high rate (56.3%) of malignant transformation after molar evacuation, primary hysterectomy was the treatment employed for the treatment of hydatidiform mole in this age group.
In our case, it was decided to perform hysterectomy because of the perimenopausal state of the patient and the presence of risk factors for post-molar GTN. Risk factors identified for post-molar GTN include age of over 40 years, an hCG level greater than 100,000 mIU/mL, excessive uterine enlargement for presumed gestational age, large theca lutein cysts (>6 cm), and a history of previous GTD. Age over 40 years and initial hCG level greater than 200,000 mIU/mL were risk factors that were present for post-molar GTN in the present case. Although suction curettage is the most frequent technique for molar evacuation, hysterectomy is an acceptable option in older patients who do not wish to preserve parity, as malignant transformation is more prone to occur in women of older age after molar evacuation (37.5% of patients aged over 50 years versus 27.5% of patients aged 40 to 49 years). Furthermore, hysterectomy decreases the risk for post-molar GTN to approximately 3.5% from the anticipated 20% following suction curettage [15]. Nevertheless, all patients should be monitored after hysterectomy because it does not completely eliminate the potential for post-molar GTN. Likewise, because of the sequential plateau of hCG lasting 3 weeks, our case was diagnosed as post-molar GTN after hysterectomy.
According to the FIGO staging system, our patient was considered as stage 3 GTN due to the presence of lung metastasis; the World Health Organization modified prognostic score was 7. Because this score was >6, the patient was treated with multi-agent chemotherapy, which includes combinations of etoposide, methotrexate, dactinomycin, cyclophosphamide, and vincristine. Our patient's serum hCG levels gradually decreased into the normal range in the second chemotherapy cycle, and two additional cycles were given in order to reduce the risk of relapse. Finally, serum hCG levels remained within the normal range (<5 mIU/mL) during follow-up 6 months after chemotherapy.
To our knowledge, this is the first case reported in the literature of a GTN presenting with uterine mass of 28 weeks' gestational size in this age group. Moreover, this case included some unusual clinical characteristics. First, this case is important in that it shows that a huge molar mass could represent invasive mole even in the absence of the invasion of adjacent organs and vascular structures, hemoperitoneum, massive bleeding, and uterine perforation. Conversely, life-threatening complications associated with massive bleeding have been reported even with smaller molar masses causing the enlargement of the uterus. Secondly, in molar diseases in perimenopausal period, abnormal bleeding of the uterus may be incorrectly perceived as menstrual irregularity in the perimenopausal period, potentially delaying diagnosis. Clinicians should consider GTNs in the differential diagnosis of patients presenting with menstrual irregularity, especially during perimenopause.
In conclusion, as an unusual cause of abdominopelvic mass, we believe that our case will contribute to the literature due to the very rare and atypical presentation of this entity during the perimenopausal period.
References
1. Ozalp SS, Telli E, Oge T, Tulunay G, Boran N, Turan T, et al. Multicenter analysis of gestational trophoblastic neoplasia in Turkey. Asian Pac J Cancer Prev 2014;15:3625-3628. PMID: 24870768.


2. Lurain JR. Gestational trophoblastic disease I: epidemiology, pathology, clinical presentation and diagnosis of gestational trophoblastic disease, and management of hydatidiform mole. Am J Obstet Gynecol 2010;203:531-539. PMID: 20728069.


3. Hol K, Junnare K, Shekhawat GS, Damle H. A case of invasive mole with uterine rupture presenting as a hemoperitoneum. Glob J Res Anal 2013;2:195-196.
4. Hurteau JA. Gestational trophoblastic disease: management of hydatidiform mole. Clin Obstet Gynecol 2003;46:557-569. PMID: 12972737.


5. Allen SD, Lim AK, Seckl MJ, Blunt DM, Mitchell AW. Radiology of gestational trophoblastic neoplasia. Clin Radiol 2006;61:301-313. PMID: 16546459.


6. In: Gibbs RS, Danforth DN, editors. Danforth's obstetrics and gynecology. 10th ed. Philadelphia (PA): Lippincott Williams & Wilkins; 2008.
7. Taskin S, Cengiz B, Ortac F. Invasive mole in a postmenopausal woman. Int J Gynaecol Obstet 2006;93:156-157. PMID: 16542659.


8. Bruner DI, Pritchard AM, Clarke J. Uterine rupture due to invasive metastatic gestational trophoblastic neoplasm. West J Emerg Med 2013;14:444-447. PMID: 24106538.



9. Kumar S, Vimala N, Mittal S. Invasive mole presenting as acute haemoperitoneum. JK Sci J Med Educ Res 2004;6:159-160.
10. Seckl MJ, Sebire NJ, Fisher RA, Golfier F, Massuger L, Sessa C, et al. Gestational trophoblastic disease: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013;24(Suppl 6):vi39-vi50. PMID: 23999759.


11. Lok CA, Zurcher AF, van der Velden J. A case of a hydatidiform mole in a 56-year-old woman. Int J Gynecol Cancer 2005;15:163-166. PMID: 15670312.


12. von Welser SF, Grube M, Ortmann O. Invasive mole in a perimenopausal woman: a case report and systematic review. Arch Gynecol Obstet 2015;292:1193-1199. PMID: 26050078.


13. de la Fouchardiere A, Cassignol A, Benkiran L, Rudigoz RC, Gougeon A, Devouassoux-Shisheboran M. Invasive hydatiform mole in a postmenopausal woman. Ann Pathol 2003;23:443-446. PMID: 14752389.

14. Tsukamoto N, Iwasaka T, Kashimura Y, Uchino H, Kashimura M, Matsuyama T. Gestational trophoblastic disease in women aged 50 or more. Gynecol Oncol 1985;20:53-61. PMID: 2981190.


15. Bandy LC, Clarke-Pearson DL, Hammond CB. Malignant potential of gestational trophoblastic disease at the extreme ages of reproductive life. Obstet Gynecol 1984;64:395-399. PMID: 6087230.

Fig. 1
(A) Giant invasive mole (28×25×15 cm) presenting with abdominopelvic mass. (B) Ultrasound images show a huge molar mass and marked thinning of the uterine wall. (C) The preoperative pulmonary computed tomography scan revealed metastatic nodules (white arrow), especially in basal segments of the right lung. (D) All of the nodules showed significant regression at 12th month after chemotherapy.
-
METRICS

-
- 8 Crossref
- 2,965 View
- 34 Download
- Related articles in Obstet Gynecol Sci