A secondary abdominal pregnancy with unusual placental implantation in the fallopian tube: a diagnostic challenge
Article information
Abstract
We reported a case of secondary abdominal pregnancy with placental implantation into the fallopian tube, diagnosed at 16 weeks, in a woman admitted to the emergency room complaining of syncopal attacks. The best approach would be termination of the pregnancy, taking into consideration the high risk to the mother and the low possibility of alive and healthy birth. We had to perform an urgent surgical intervention due to the fact that the patient was in a clinically unstable condition, which was related to hemoperitoneum. If placental implantation is on abdominal organs or vessel the best approach would be to ligate the cord and to leave placenta in situ. Taking into consideration the place of placental implant, the removal of the fallopian tube with the placenta was the safest approach in this case. The best and most acceptable form of treatment would be individualized in case of rare form of ectopic pregnancy.
Introduction
In more than 95% of cases, ectopic pregnancies occur in the fallopian tube. In the remaining cases, the pregnancy development is cervical, interstitial (or cornual), on hysterotomy (cesarean) scar, intramural, ovarian, or abdominal [1].
Abdominal pregnancy accounts for up to 1.4% of ectopic pregnancies and could result from a secondary implantation of an aborted tubal pregnancy or from an intraabdominal fertilization with primary implantation in the abdomen [23].
Implantation sites, external to the uterine cavity and fallopian tubes, include the omentum, the pelvic sidewall, the broad ligament, the posterior cul-de-sac, the spleen, the bowel, the liver, the large pelvic vessels, the diaphragm, and the uterine serosa [2]. Risk factors for abdominal pregnancy include tubal damage, pelvic inflammatory disease, endometriosis, assisted reproductive techniques and multiparity [4]. Because of the variable location in the abdomen, a wide range of signs and symptoms are described. Considering that this condition may go undetected until advanced gestational age, occasionally within the term, it often results in severe hemorrhage, so maternal mortality rate could be as high as 20% [356]. The best clinical practice is the termination of abdominal pregnancy at the time of diagnosis, even in the case of advanced gestational age. In fact, the risk of life threating maternal complications is too high and the possibility of the delivery of a healthy infant is low; in fact, fetal deformations (such as facial/cranial asymmetry, joint abnormalities, hypoplastic limbs, central nervous system malformation) and perinatal death occur frequently [78]. Expectant management to gain the fetal maturity has been attempted and has been successful in a few cases, however adequate counselling must be offered to parents and a very close monitoring of the pregnancy is necessary [9]. If the diagnosis is in the first trimester, laparoscopy is an option; however, this treatment must be avoided in case of involvement of vascular surface [10]. Methotrexate therapy has a low success rate, possibly due to advanced gestational age at the time of diagnosis [11]. Considering the high risk of life threatening maternal hemorrhage, ligation of the umbilical cord and leaving the placenta in situ could be considered a safe option [121314]. An alternative approach would be to ligate the placental blood supply and then try to remove the placenta. However, this is generally considered to be a difficult procedure, mostly in case of implantation on vital organs or large blood vessels [34]. Preoperative selective arterial embolization may also help to prevent hemorrhage during attempts to remove the placenta [1213].
Case report
A 34 years old woman was admitted to our emergency room, brought in by ambulance; she complained of 3 syncopal attacks at home as well as an abdominal pain lasting over a period of 2 days.
In her medical history, she did not report any previous disease or surgery. She had both a 4 and a 2 years old child, who had been vaginally delivered with no complications. She had never used any contraceptive and did not have any history of pelvic inflammatory disease. She only suffered from oligomenorrhea and her last menstrual period was unknown.
When she arrived in the emergency ward, the patient was severely pale. Her pulse rate was 100/minute and her blood pressure was 125/80 mmHg.
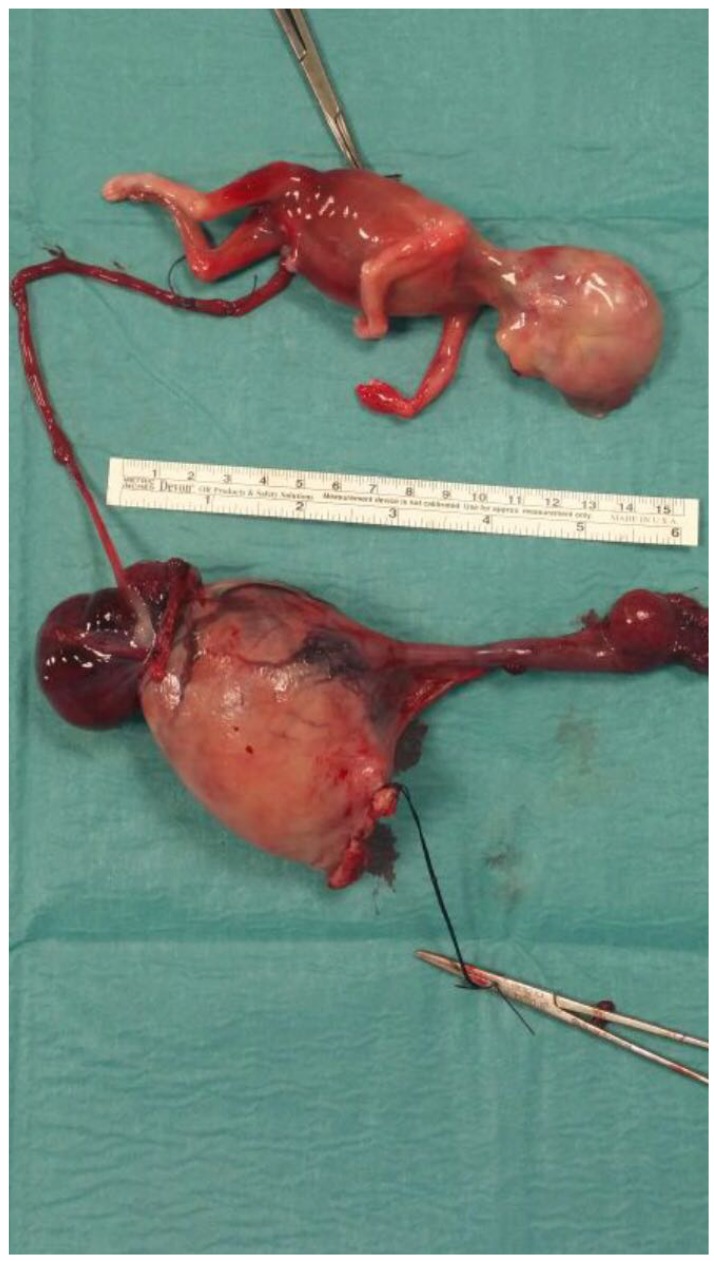
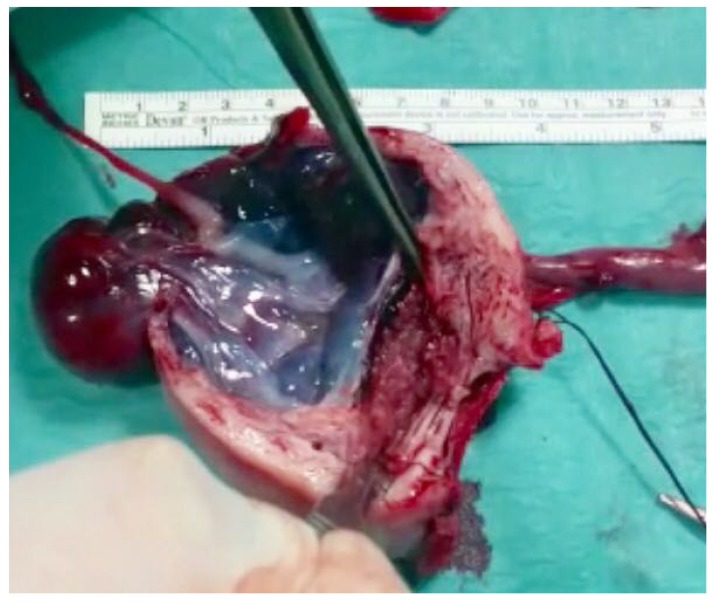
Abdominal assessment appeared slightly tender. No particular pain was evocated and no masses were found. The examination was limited to patient's elevated body mass index of 40. Bi-manual vaginal examination revealed a normal uterine size and a palpable and painful left adnexal mass; the right ovary was not detected. Digital palpation of the punch of Douglas was deeply painful. No vaginal bleeding was observed. At vaginal and abdominal ultrasound examination (Fig. 1), we saw an anti-verted uterus of 97×57 mm, with decidual reaction, a left adnexa with a hyperechoic mass of 68×56 mm, with no univocal interpretation and a normal right ovary of 32×13 mm. There was a moderate amount of free fluid in the pelvis, in the Morrison's pouch and in the paracolic gutters. We also identified a rounded image with hyperechoic boundaries, which led us to believe that there was a suspicion of a fetal head in the upper right side of abdomen, which was difficult to study taking into consideration also the bowel movements and the patient's habitus. The woman did not know her eventual obstetric status. Thanks to blood tests, we were able to confirm our hypothesis: beta human chorionic gonadotropin (hCG) was 78,079 mIU/mL; other investigations were hemoglobin of 8.4 g/dL, platelets 333,000/µL, and normal coagulation parameters. An abdominal radiography was required in order to confirm the fetal presence and its location; however, it did not identify fetal bones. During recovery, she had a further syncopal attack, in spite of an infusion of intravenous fluids, with blood pressure of 80/40 mmHg, in addition to a feeble and tachycardic pulse rate. This clinical status aggravation required an urgent surgical intervention, which meant that a longitudinal laparotomy was performed. At celiotomy, we found one liter of blood with many clots and we discovered in the left paracolic gutter a dead fetus showing a development about 16 weeks. The fetus was lying freely in the peritoneal cavity and the umbilical cord connected him to the anti-mesial surface of the left fallopian tube, which appeared enlarged to about 10 cm. We suspected that the placenta's implantation was inside the adnexa. The anti-mesial surface showed continuous dripping, so we removed the enlarged tube with the fetus connected by the umbilical cord (Fig. 2). The surgical intervention was concluded with an accurate hemostasis and the abdomen closure. An intraoperative macroscopic examination, after a longitudinal left tube cut, confirmed our hypothesis: the placenta was located into the adnexa (Fig. 3). During the procedure, the patient was transfused with 3 units of whole blood and 2 units of fresh frozen plasma. Post-operative period was uneventful. At discharge, 4 days after, the beta hCG value dropped to 586 mIU/mL, hemoglobin value was 10.1 g/dL and the ultrasound check was normal. After 2 weeks, a follow up check was performed, showing an overall stability in her health and a decrease in her beta hCG.
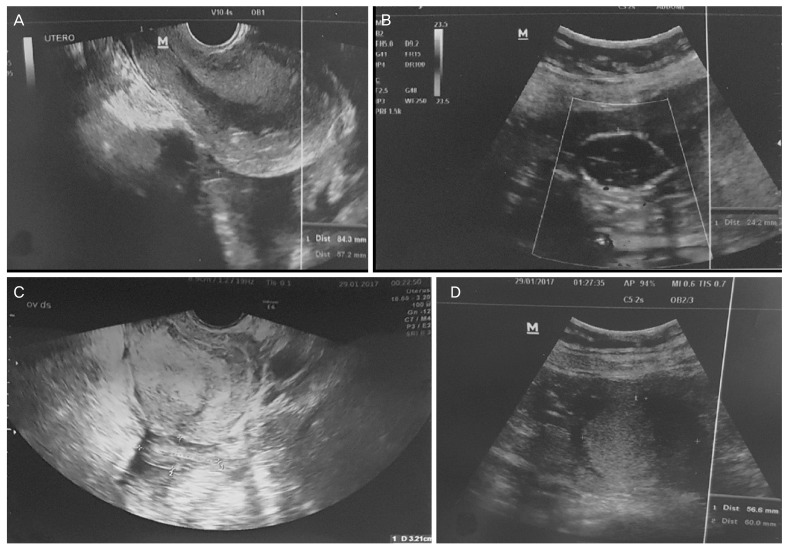
Ultrasound finding at the fist evaluation at emergency room. (A) the anti-verted empty uterus; (B) a rounded image with hyperechoic boundaries, suggestive of fetal head in the upper right side of abdomen; (C) a normal right ovary of 32×13 mm; (D) a left adnexa with a hyperechoic mass of 68×56 mm, with not univocal interpretation.
The patient signed a form of consent for the use of images and videos taken of during the procedure. Institutional Review Board approval was not required for this kind of treatment.
Discussion
Abdominal pregnancy is a rare condition, which must be suspected in case of ectopic pregnancy and requires a prompt treatment, considering the high maternal risk. The approach must be individualized. The difficulty in these clinical conditions is correlated with their rarity and variability.
In literature, 51 cases, resumed in Table 1, have been reported from January 2013, and Supplementary Table 1 described more detailed data of these cases. Twenty-two cases were primary abdominal pregnancy. Various modality of diagnosis and medical or surgical choice were performed in different cases. Specifically, ultrasound could be helpful for the diagnosis, but there are different cases in which the ultrasound evaluation erroneously described a intra uterine pregnancy. Placental implantation was in different places: uterine external wall, adnexa, broad ligament, bowel, and omentum were the most frequent implantation sites; but also spleen, hepatic surface and sacral plexus were interested in some cases. The case we described is a rare condition of secondary abdominal pregnancy. In fact, our patient presented a fetus in the abdomen with its placenta attached to the fallopian tube. To the best of our knowledge, only one similar case has ever been presented in literature [15].
Laparotomy was the more frequent treatment in these cases. Laparoscopy was reported in 11 cases diagnosed in the first trimester. In some situations, methotrexate was used, but only in one case, associated with potassium chloride this therapy was conclusive. A case of maternal death, correlated to misdiagnosis, happened. In one case, an expectant management was performed, considering the not viable gestational age of the fetus at diagnosis, but a fetal demise happened. Eleven neonates were alive (different period of follow up was reported) and in one case of these cases dimorphisms were described.
Our patient presented only multiparity as a risk factor [4]. As usually occurs, the pregnancy was undetected upon arrival in the emergency room, and then the diagnosis was performed during the patient evaluation for syncopal attacks. In the case of a confused/unconscious women, with abdominal pain or vaginal bleeding, at fertility age, the evaluation must provide the evaluation of hemodynamic stability/instability, the exclusion/confirmation of a pregnancy (and its eventual localization); additional testing could be essential guides for further management throughout these cases. Specifically, the diagnosis of pregnancy could be done thanks to clinical evaluation, ultrasound examination and measurement of beta hCG. Magnetic resonance could be useful in unclear situation and cases, but this cannot be attempted with women showing hemodynamic instability. Our suggestion for diagnosis in the case of hemodynamically stable patients was to perform a computed tomography or magnetic resonance because of the difficulties in identifying the pregnancy using only clinical evaluation and ultrasound examination. In fact, as reported in literature (Table 1), ultrasounds have not always been conclusive and sometimes even confusing, and other evaluations could be required or the diagnosis could be done at the time of cesarean section. Considering the inability to perform a magnetic resonance in our hospital during the night, we decided to require an abdominal radiography, but it was not useful, because fetal bones are probably very difficult to see with this type of investigation, so we did not suggest to perform X-ray in case of diagnostic doubts. When patient presented hemodynamic instability the surgical evaluation is, for us, the best option as it is a diagnostic and curative approach.
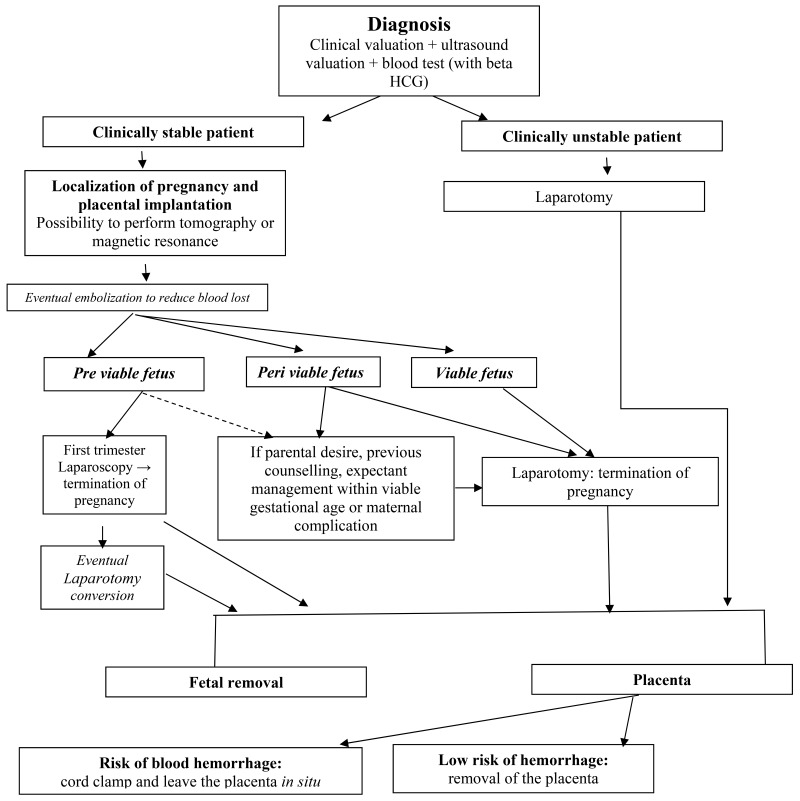
Appendix 1 summarized, thanks to a diagram, a possible clinical management. Specifically, when the diagnosis was defined, we suggested the termination of the pregnancy in case of hemodynamic instability; in case of clinical stable and asymptomatic patient, with an accidental diagnosis, we would evaluate the gestational age. In case of pre-viable fetus we would suggest pregnancy termination and we would underline the possibility of laparoscopic management in case of diagnosis in the first trimester. If gestational age was peri-viable, then, pregnancy termination would be the best choice, taking into consideration the high maternal risk and the low chance of a healthy fetus, then a laparotomy must be performed. If the fetus was at viable gestational age, the procedure advised would be abdominal delivery. In case of parental desire of expectant management adequate counselling, clarifying maternal risk and poor fetal condition, must be offered to the patient. However, in literature interruption of pregnancy, thanks to surgery, methotrexate or potassium chloride, is the best practice also in the case of incidental diagnosis in asymptomatic patient [78].
When termination of the pregnancy has been decided and agreed upon by the parents then a surgical approach must be individualized. We recommended the removal of the placental implantation site in case of the organ was not indispensable for life. In literature, a high risk of life threatening maternal hemorrhage was described in the case of placental removal and ligation of umbilical cord and therefore, the leaving of the placenta in situ could be a safe option. In these cases, the placenta in situ could leading to abdominal pain or fever due to necrosis or infection [121314]. Preoperative selective arterial embolization was reported to prevent hemorrhage during placenta removal, but in case of patient hemodynamic instability, this procedure cannot be performed [1213].
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Appendix
Appendix 1
Diagram resumed the therapeutic algorithm for the abdominal pregnancy
References
Supplementary Material
Supplementary Table 1
Cases of spontaneous abdominal pregnancies reported in literature (English publications), from January 2013 to May 2017, compared to our case