Hemoperitoneum: a complication of loop electrosurgical excision procedure
Article information
Abstract
Loop electrosurgical excision procedure (LEEP) is commonly performed for the management of cervical intraepithelial neoplasia. Although LEEP is considered to be a relatively simple procedure, several unexpected complications have been reported in the literature. Herein, we report a case of hemoperitoneum caused by uterine perforation following LEEP. Blood collection in pelvic cavity and two small defects of the uterus were confirmed by diagnostic laparoscopy. The defects were sutured and the patient recovered well after the operation.
Introduction
Loop electrosurgical excision procedure (LEEP) is the preferred management for a high-grade squamous intraepithelial lesion (HSIL). Common complications of LEEP are vaginal bleeding, preterm delivery, and cervical stenosis [1]. Unusual complications of LEEP are postoperative peritonitis, vesicovaginal fistula, vaginal evisceration, and lower urinary tract injury [2345]. Hemoperitoneum is a rare complication after LEEP, according to the available literature. Herein, we present a case of hemoperitoneum following LEEP.
Case report
A 27-year-old woman, gravida 1 para 1, was referred for the management of HSIL. LEEP was performed uneventfully under monitored anesthesia care using propofol. A 2.4×2.8×0.8 cm sized specimen was obtained after multiple passes with 1.5×1.5 cm sized electrodes. A little bleeding that was observed at the 6-o'clock position on the cervix was electrocoagulated and sutured with Vicryl 2-0®(Ethicon, Somerville, NJ, USA), encircling the posterior part of the cervical os. Then, a piece of gauze was packed in vagina for compression.
An hour after operation, she experienced upper abdominal pain. We removed the gauze and examined her cervix, but the findings were nonspecific. Two hours after operation, her blood pressure measured 84/43 mmHg and pulse rate was 82 beats per minute. She responded well to intravenous fluid resuscitation and the blood pressure recovered to 108/63 mmHg, and the pulse to 73 beats per minute. Blood tests revealed a reduced hemoglobin level of 10.6 g/dL from the preoperative hemoglobin level of 12.0 g/dL. Her abdominal pain was relieved after defecation, but she complained of persisting bilateral subcostal pain. Postoperative radiographs of the abdomen and chest did not reveal any concerning findings. Next morning, her hemoglobin level had further reduced to 9.2 g/dL. As no vaginal bleeding had occurred postoperatively, this reduction could not be attributed much to hemodilution.
Typically, patients undergoing LEEP in our hospital are discharged one day after the procedure. We postponed discharge because her clinical course was not usual. Abdominal and pelvic contrast-enhanced computed tomography (CT) was performed to assess the cause of her abdominal discomfort. Imaging studies revealed a moderate amount of blood collection in the pelvic cavity and no observable active bleeding site (Fig. 1). Ectopic pregnancy was excluded as her beta human chorionic gonadotropin levels were less than 0.100 mIU/mL. Transvaginal ultrasonography found fluid collection in the posterior cul-de-sac and no abnormal findings in the uterus and adnexa.
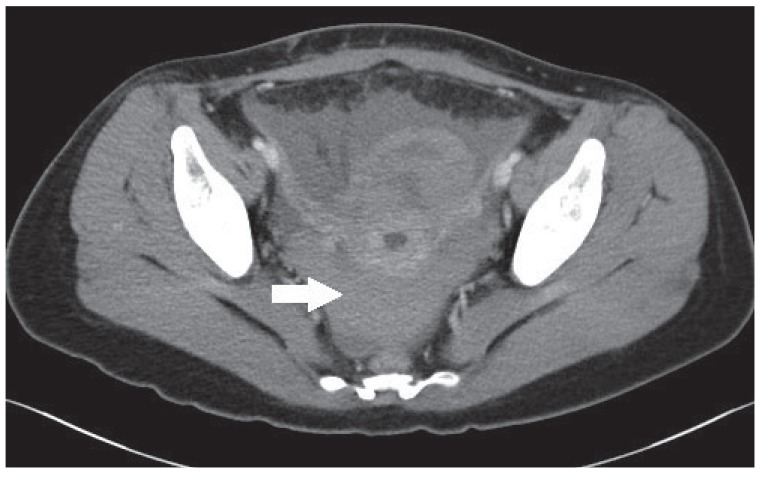
Abdominal and pelvic contrast-enhanced computed tomography showing fluid collection in pelvic cavity (arrow).
To pinpoint the origin of the bleeding site and the cause of hemoperitoneum, diagnostic laparoscopy was performed. A uterine manipulator was not used during operation because her cervix was friable due to LEEP. On entering the peritoneal cavity, approximately 500 mL of old blood was noted (Fig. 2A). After removal of the blood collection, the abdominopelvic cavity was thoroughly inspected. There were two 0.5 cm sized symmetric defects above both uterosacral ligaments without active bleeding (Fig. 2B). Since there was no other abnormal finding in abdominopelvic cavity, we suspected that these defects had caused the hemoperitoneum. After the defects were sutured laparoscopically using Vicryl 2-0®(Ethicon), a diagnostic cystoscopy was performed. Indigo carmine was injected intravenously and excretion of dyed urine through both ureteral orifices was observed. The postoperative course was uneventful, and she was discharged on the third postoperative day.
Discussion
LEEP is a common procedure that is performed in cases of cervical intraepithelial neoplasia (CIN) using a small wire loop and electric current [6]. Common complications of LEEP are vaginal bleeding, infection, cervical stenosis, and cervical incompetence [17]. Peritonitis, vesicovaginal fistula, vaginal evisceration, and lower urinary tract injury have been reported as unusual complications [2345]. To avoid iatrogenic injury during LEEP, profound understanding of anatomy is necessary.
The cervix of the woman of reproductive age measures 3.5 cm in length and 2 cm in diameter [8]. After menopause, atrophic changes of cervix are common. Merz at al. [9] reported that the mean cervical length of women who had been postmenopausal at least 5 years was 2.4 cm. Therefore, the use of extremely large electrodes (e.g., 20×20 mm) may lead to injury of the vaginal walls, rectum, and bladder [7]. If the diameter of a lesion is larger than 2 cm, the lesion must be removed with multiple passes using one or more sizes of loop [10]. If there is marked atrophy due to estrogen deficiency in older women, it is advisable to delay LEEP until after a course of topical estrogen treatment [10].
Several studies have shown that the endocervical mucosa projects 3 mm into the underlying stroma but may plunge as deep as 6 mm [8]. CIN can penetrate the stroma a depth of 3 to 3.5 mm and rarely to a depth of up to 6 mm [11]. Therefore, if the lesion involves the endocervical canal, the depth of excision should be at least 6 mm. The preferred depth of the excision is 7 to 8 mm, and a “top-hat” technique can be used for deep endocervical lesions [7]. As the depth of excision increases, the risk of bleeding and stenosis increases [10]. For this reason, if the lesion extends more than 1 cm into the canal, LEEP should not be used [10].
We suspect that the hemoperitoneum in our case was caused by uterine perforation. Unintentional colpotomy during the procedure may have occurred during electrocoagulation or suturing for hemostasis. Several risk factors of iatrogenic injury during LEEP have been reported in the literature. Moon et al. [12] reported that deep suturing during conization resulted in the formation of a hematoma adjacent to the uterine artery. Ghassani et al. [4] reported that the use of monopolar coagulation in an atrophic cervix caused iatrogenic colpotomy during LEEP. Cho et al. [3] suggested that atrophic cervix and endocervical lesion are risk factors of over-extended excision of cervix and injury of adjacent tissue. In addition, the placement of the suture through both anterior and posterior lips of cervix before the procedure may have enabled a precise cut of the cervix and reduced complications [13].
We identified only one case report of abdominal bleeding following LEEP by searching in PubMed [14]. Since hemoperitoneum is uncommon after LEEP, it is easy to be overlooked. Furthermore, LEEP is usually performed at the outpatient clinic and complications except vaginal bleeding can be easily neglected. Our case illustrates the difficulties in early diagnosis of hemoperitoneum following LEEP. Moreover, when the patient is hemodynamically stable, the physician may be unaware of the presence of hemoperitoneum. Similarly, a minor hemoperitoneum after LEEP can be underreported.
In our case, we confirmed the defects of the uterus by laparoscopy. Uterine perforation can cause bowel perforation or peritonitis as well as hemoperitoneum [212]. Therefore, when a patient complains of abdominal pain after LEEP, physicians should postpone the discharge and observe the clinical course of the patient carefully. In addition, ultrasonography or CT can be useful diagnostic tools for a patient with unexplained abdominal pain after LEEP.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Patient consent: The patients provided written informed consent for the publication and the use of their images.

