Fetal cardiac function by mitral and tricuspid annular plane systolic excursion using spatio-temporal image correlation M-mode and left cardiac output in fetuses of pregestational diabetic mothers
Article information
Abstract
Objective
To assess the mitral and tricuspid annular plane systolic excursions (MAPSE and TAPSE, respectively) and cardiac output (CO) in fetuses of pregnant women with pregestational diabetes mellitus (DM) using spatio-temporal image correlation M-mode (STIC-M) and virtual organ computer-aided analysis (VOCAL).
Methods
This study was prospective and cross-sectional. It included 45 fetuses each from mothers with pregestational DM and healthy mothers, with gestation ages ranging from 20 to 36.6 weeks. The fetal cardiac volumes were obtained and analyzed by STIC and VOCAL methods. MAPSE and TAPSE were measured by STIC-M in the apical or basal four-chamber view. The values of the right (RV) and left ventricular (LV) CO were calculated by STIC and VOCAL.
Results
The median values of TAPSE were 6.1 and 6.2 mm in the diabetic and control groups (P<0.001), respectively. The median values of MAPSE were 4.6 mm in the fetuses of mothers with diabetes and 4.8 mm in fetuses of healthy mothers. The fetal LV CO (60.4 L/min vs. 71.1 L/min; P=0.033, respectively) and RV CO (65.2 vs. 70.1 L/min; P=0.026, respectively) were lower in the pregestational DM group than in the control group. A significant effect of pregestational DM was observed in all functional parameters after adjusting, with fetal heart rate as covariant. There was moderate significant positive correlation between MAPSE and LV CO (r=0.53; P=0.0001) and between TAPSE and RV CO (r=0.46; P=0.0001).
Conclusion
Significant difference in functional parameters (TAPSE, MAPSE and LV CO) obtained by STIC and VOCAL were observed in the fetuses of the pregestational DM group compared to those of the control group.
Introduction
Classically, gestational diabetes mellitus (DM) is the main cause of cardiac hypertrophy in fetuses. Pregestational DM affects structural and functional cardiogenesis, especially when the maternal blood glucose is uncontrolled [1–4]. Studies have demonstrated that fetuses of diabetic mothers may present signs of cardiac dysfunction even in the absence of septal hypertrophy [5,6]. It is, therefore, important to assess the functional parameters of ultrasound/echocardiography to detect subclinical cardiac dysfunction in this population [7,8].
Traditionally, the global cardiac systolic function can be assessed by shortening fraction (SF) using two-dimensional (2D) ultrasound or by ejection fraction (EF) using three-(3D) and four-(4D) dimensional ultrasound with spatio-temporal image correlation (STIC) and virtual organ computer-aided analysis (VOCAL) methods [9,10]. SF is applied to evaluate the global radial contractility by measuring the reduction of the ventricular diameter of end-diastole to end-systole [11,12]. Using advanced techniques such as 3D ultrasound, the EF reflects the percentage of blood ejected by the ventricles in each cardiac cycle and can be used to evaluate the longitudinal myocardial function with good reproducibility [10,13,14].
The longitudinal cardiac contractility can be assessed by measuring the maximum mitral and tricuspid annular plane systolic excursions (MAPSE and TAPSE, respectively). These parameters can be affected in the early stages of cardiac dysfunction and can be obtained by means of ultrasound using conventional anatomical M-mode tracings and by STIC M-mode (STIC-M) [15]. Studies on fetal TAPSE and MAPSE have demonstrated the applicability of these parameters on risk factors for fetal heart failure, such as fetal growth restriction and maternal DM [16–18].
The aim of this study was to compare the values of fetal MAPSE, TAPSE, and cardiac output (CO) obtained by STIC-M and VOCAL in fetuses from pregnant women with DM and controls.
Materials and methods
This study was prospective and cross-sectional, performed to evaluate fetal MAPSE, TAPSE, and CO in fetuses from diabetic mothers compared with those in fetuses from low-risk pregnant women between 20 and 36.6 weeks of gestation. From November 2016 to November 2018, all data underwent routine ultrasound evaluation and fetal echocardiogram at the same time at the Fetal Cardiology Sector of the Federal University of São Paulo (UNIFESP).
The DM and control groups included fetuses of pregnant women with pregestational DM types 1, 2, or maturity-onset diabetes of the young and those of healthy mothers, respectively. The inclusion criteria were as follows: singleton pregnancies with gestational age (GA) between 20 and 36.6 weeks determined by the last menstrual period and confirmed by first trimester ultrasound. The exclusion criteria were as follows: fetuses whose mothers had other comorbidities except pregestational DM; estimated fetal weight <10th percentile for GA; smokers or drug/alcohol users; fetuses with structural cardiac and extracardiac anomalies at ultrasound examination; and fetal cardiac volumes with inadequate quality images due to fetal movements, presence of acoustic shadows of fetal ribs or spine, and maternal breathing.
Pregnant women were assessed once. The obstetrical ultrasound and fetal echocardiogram examinations were performed on the same day using a Voluson E6 apparatus (General Electric Healthcare, Zipf, Austria) equipped with 3.0–5.0 MHz convex volumetric probe. Examinations were performed by two examiners (NJMBV and ABP) with experience in both 3D ultrasound and fetal echocardiography. First, evaluation of the fetal morphology and biometry were assessed by 2D ultrasound. Fetal cardiac volumes were acquired in the four-chamber view according to Gonçalves et al. [19]. The angle of the scan acquisition was standardized, ranging from 20º to 25º and from 35º to 40º in the second and third trimesters, respectively, with a speed time ranging from 7.5 to 15 seconds. One to three fetal cardiac volumes were obtained from each pregnant woman and stored in DICOM. The off-line analysis was performed by the principal investigator (NJMBV) using 4D view software version 10.0 (General Electric Medical System, Zipf, Austria). The axial plane (A) of the multiplanar mode was selected, and the fetal heart was rotated around the ‘z’ axis for the fetal spine to be at a 6 o’clock position. Image adjustments with sepia or candle colors and cineloop mode were reduced to 6%. To measure fetal MAPSE and TAPSE, the STIC-M key was activated and the cursor was placed at the atrioventricular junction, marked by mitral and tricuspid valves, respectively. The maximum amplitude of motion was obtained as per the extent of displacement between end-systole and end-diastole (measured in mm) (Fig. 1). The maximal and minimal ventricular volumes were measured at the end-diastole (just after closing of the atrioventricular valves) and end-systole (before opening of the atrioventricular valves), respectively. Subsequently, the caliper was positioned in the atrioventricular valve and in the ventricular apical region (Fig. 2). Then, the VOCAL key was activated, and this software provided the reconstruction of the 3D image of each ventricle after 6 sequential manual planes (30°). The EF and CO for left and right ventricles were calculated using the following formulas: EF=end diastolic ventricular volume–end systolic ventricular volume/end diastolic ventricular volume and CO=end diastolic ventricular volume–end systolic ventricular volume×heart rate.

Measurement of the fetal left ventricle (LV) volume using spatio-temporal image correlation and virtual organ computer-aided analysis (VOCAL) methods in a fetus from pregestational diabetes mellitus at 22 weeks and 5 days of gestation (LV diastolic volume=0.48 cm3 and heart rate [HR]=166 bpm). The LV cardiac output (LV CO) and LV ejection fraction (LVEF) were calculated from the measurements of the ventricular volumes according to the following formulas: LV CO=(LVDv−LVSv)×HR and LVEF (LVDv−LVSv)/LVDv. The calipers were positioned on the internal border of the mitral valve and on the ventricular endocardial border to perform the manual delimitation of the internal left ventricular volume. After 6 sequential planes, the VOCAL software provided the chamber volume and the reconstructed 3D image. LVDv, left diastolic volume; LVSv, left systolic volume.
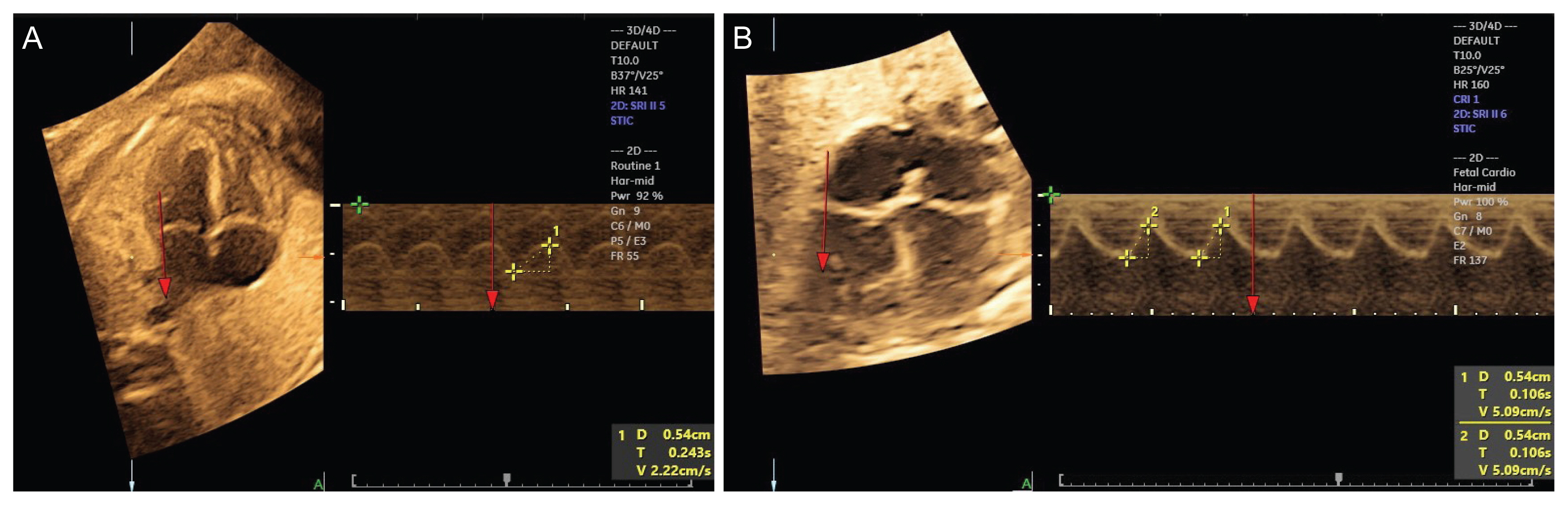
In the apical four-chamber view of the fetal heart, the spatio-temporal image correlation in M-mode image is positioned at the junction between the free ventricular wall and the atrioventricular valve for the calculation of mitral annular plane systolic excursion (MAPSE) (A) and tricuspid annular plane systolic excursion (TAPSE) (B) annular plane systolic excursions. MAPSE=5.4 mm; TAPSE=5.4 mm.
The following clinical variables were also collected to analyze the characteristics of the study population: maternal age, number of previous pregnancies, parity, number of miscarriages, GA at the time of ultrasound examination, estimated fetal weight, fetal heart rate (FHR), and last serum glycated hemoglobin levels.
Data were transferred to an Excel 2010 spreadsheet (Microsoft Corp., Redmond, WA, USA) and analyzed using PASW version 20.0 (SPSS Inc., Chicago, IL, USA) and GraphPad (version 7.0, GraphPad Software, San Diego, CA, USA). Continuous variables were expressed as the medians (minimum and maximum ranges). Categorical variables were expressed as percentages. The Mann-Whitney U-test was used to compare the two groups, with a critical alpha of 0.05. Spearman’s correlation test (r) was used to determine the correlation between continuous variables. Linear regression was used to analyze the influence of the FHR on fetal cardiac function parameters, the influence of MAPSE on the left ventricle (LV) CO, and the influence of TAPSE on the right ventricle (RV) CO. General linear model with FHR as covariant was used to evaluate the influence of pregestational DM on fetal cardiac parameters.
Results
We evaluated 104 pregnant women. However, 13 fetuses were excluded because of the poor quality of the fetal cardiac volume acquisition, 1 case because the estimated fetal weight was <10th percentile for GA, and 2 cases because of fetal structural anomalies. Consequently, we evaluated a total of 90 fetal cardiac volumes, which were divided in two groups (45 fetuses each from mothers with pregestational DM and healthy mothers).
There was a significant effect of pregestational diabetes on maternal age (P=0.004), number of previous pregnancies (P<0.01), parity (P=0.005), and number of miscarriages (P=0.001). Healthy pregnant women had higher maternal age (33.0 vs. 27.0 years, P=0.004) and lower number of previous pregnancies (1.0 vs. 2.0, P<0.001) than pregnant women with pregestational DM (Table 1).
Considering all cases included in the study, there was a weak significant negative correlation between FHR and the following: TAPSE (r=−0.37, P=0.0003), MAPSE (r=−0.31, P=0.0029), LV CO (r=−0.29, P=0.0049), and RV CO (r=−0.30, P=0.0035) (Table 2). The elevation of 1 beat per minute (bpm) in the FHR was responsible for reducing TAPSE by 0.04 mm, MAPSE 0.03 mm, LV CO 0.79 L/min, and RV CO 0.94 L/min. According to the coefficient of determination (R2), 15%, 12%, 5%, and 6% of the variations in TAPSE, MAPSE, LV CO, and RV CO, respectively, were linearly related to FHR, with the remaining variations resulting from other factors not considered in the model (Table 2 and Fig. 3). Considering all cases of mothers with pregestational DM, whose glycated hemoglobin (HB) (%) measurements had been performed, there was a weak significant negative correlation with TAPSE (r=−0.37, P=0.019) and MAPSE (r=−0.38, P=0.015). There was not significant correlation between glycated HB (%) and LV CO (r=0.04, P=0.783) and RV CO (r=0.03, P=0.819) (Table 3).
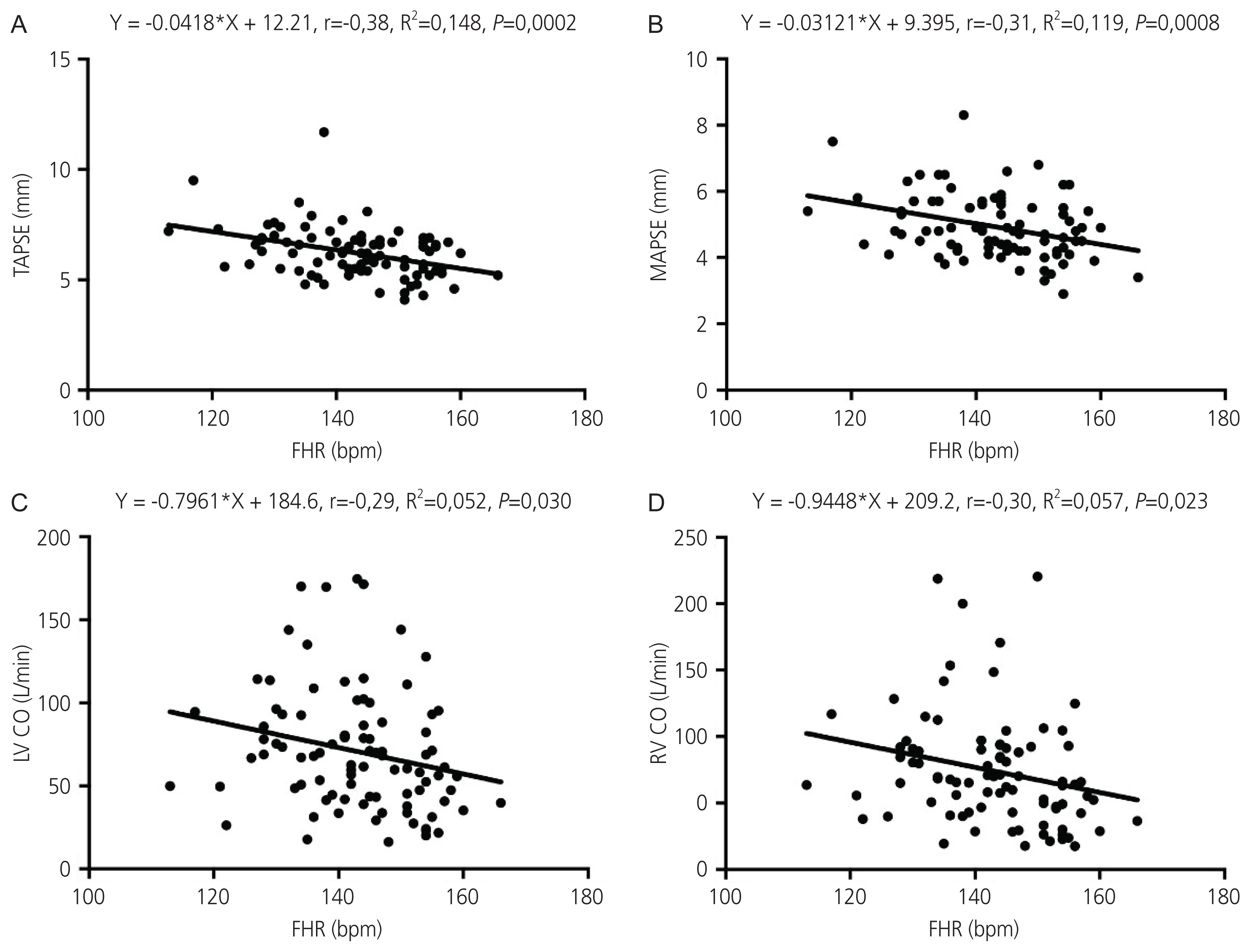
Scatter plot of the correlation between tricuspid annular plane systolic excursion (TAPSE) (A), mitral annular plane systolic excursion (MAPSE) (B), left ventricle cardiac output (LV CO) (C), right ventricle cardiac output (RV CO) (D), and fetal heart rate (FHR).
There was a significant effect of pregestational DM on TAPSE (P<0.001), MAPSE (P<0.001), LV CO (P=0.033), and RV CO (P=0.026) after adjusting, with FHR as covariant. The median TAPSE (6.1 vs. 6.2 mm, P<0.001, respectively), MAPSE (4.6 vs. 4.8 mm, P<0.001, respectively), LV CO (60.4 vs. 71.1 L/min, P=0.033, respectively), and RV CO (65.2 vs. 70.1 L/min, P=0.026, respectively) were lower in the pregestational DM group than in the control group (Table 4).
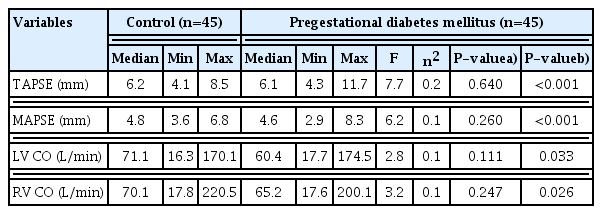
Fetal cardiac functional parameters by spatio-temporal image correlation in M-mode and virtual organ computer-aided analysis
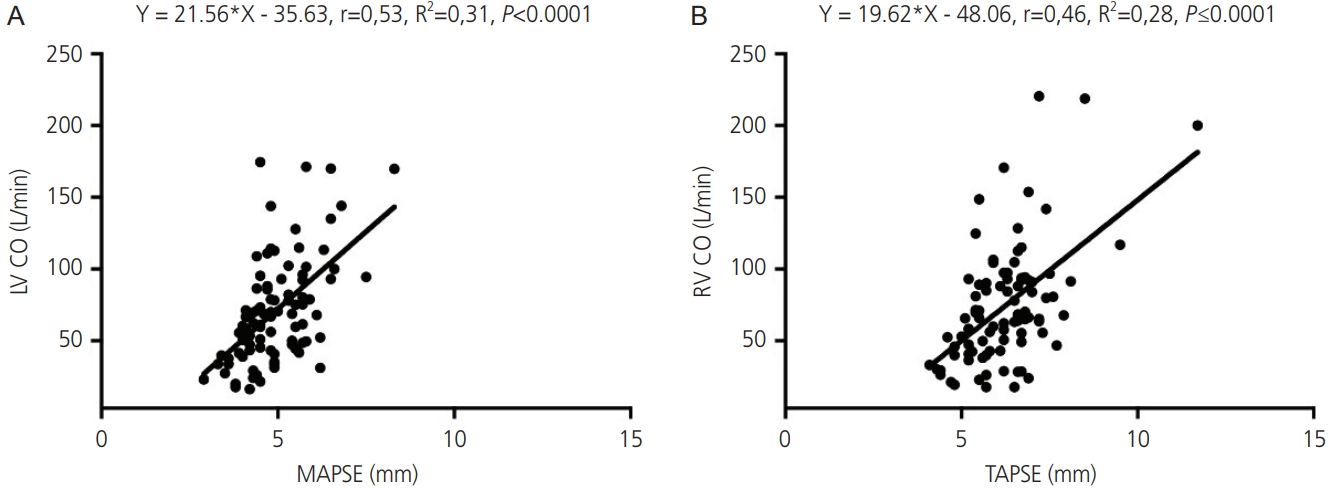
Considering all included cases, there was moderate significant positive correlation between MAPSE and LV CO (r=0.53, P<0.0001) and TAPSE (r=0.46, P<0.0001) and RV CO (Table 5). The 1-mm increase in the excursion of LV annulus was responsible for increasing the LV CO by 21.56 L/min and RV CO by 19.62 L/min. According to the R2, 31% and 28% of the variations in LV CO and RV CO were linearly related to MAPSE and TAPSE, respectively, with the remaining variations resulting from other factors not considered in the model (Table 5 and Fig. 4).
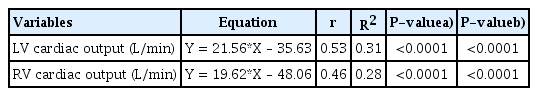
Influence of tricuspid annular plane systolic excursion on RV cardiac output and influence of mitral annular plane systolic excursion on LV cardiac output
Discussion
TAPSE and MAPSE measurements can be obtained either by 2D ultrasound using anatomical M-mode tracings or by advanced methods such as 3D/4D ultrasound with STIC (STIC-M) [20]. Traditionally, it was demonstrated that the conventional 2D M-mode is a simple and useful technique for the assessment of the longitudinal systolic cardiac function in fetuses. Fetal reference ranges for this parameter were established [20–23]. Nevertheless, some authors have demonstrated the advantages of the STIC-M over the conventional M-mode. The 3D/4D ultrasound STIC enables the optimization of the post-processed images by rotating the fetal cardiac volumes, minimizing limitations addressed to an unfavorable fetal position, and increasing the accuracy of the TAPSE and MAPSE measurements [17,24]. Moreover, other functional parameters such as CO and EF, which can be obtained with good reproducibility using 3D/4D ultrasound methods and reference curves, have been validated [13,25–27].
Messing et al. [17] demonstrated that it was possible to obtain the measurements of fetal TAPSE by 3D ultrasound with STIC, and their measurement values were similar to those obtained by the anatomical 2D M-mode. These authors included a large sample size and provided the first reference ranges of fetal TAPSE by STIC with 341 low-risk singleton pregnant women. They demonstrated that the STIC M-mode fetal TAPSE increased linearly with the GA, with mean values ranging from 4.2 mm at 21 to 8.8 mm at 39 weeks of gestation. In our study, we observed a positive correlation between fetal TAPSE and the GA, with a mean of 4.1 mm at 20 and 8.5 mm at 36 weeks of gestation in healthy fetuses.
Tedesco et al. [18] determined reference ranges for fetal TAPSE using STIC-M in 300 fetuses between 20 and 39 weeks of gestation and observed linear increase with GA. These authors observed that the fetal TAPSE values were lower in fetuses of mothers with pregestational DM than in fetuses of healthy mothers. Furthermore, in Tedesco’s study, the parameter used to assess the global systolic cardiac function was the SF obtained by STIC-M. Significant differences were observed between the two groups. Similarly, in this study, the median values of fetal TAPSE by STIC-M were lower in fetuses of mothers with pregestational DM than in fetuses of healthy pregnant women. In our study, the global systolic cardiac evaluation was assessed by CO using STIC and VOCAL. A significant effect of pregestational DM on the measurements of RV and LV CO was observed after adjusting, with FHR as a covariant.
Interestingly, in other cross-sectional studies from our group using STIC and VOCAL, no significant differences in CO and EF were observed between the fetuses of pregnant women with pregestational DM and the fetuses of healthy mothers. However, significant differences in fetal left atrial volumes between the two groups were observed. This could be related to left atrial dysfunction, reflecting an earlier stage of cardiac dysfunction (left heart diastolic dysfunction) [28]. Alternatively, in this study, the LV and RV CO were lower in fetuses of those in the pregestational DM group than in the fetuses of those in the control group. We believe that this difference could be related to the inclusion of pregnant women with gestation periods of up to 36 weeks.
Since 2004, various studies showed that the CO calculated using 3D/4D ultrasound was positively correlated to the GA [13,27,29]. Accordingly, we observed an increase in the RV CO and LV CO values with the GA. Furthermore, the parameters that we used to assess the longitudinal and radial cardiac functions were correlated positively (TAPSE and MAPSE with RV CO and LV CO, respectively).
MAPSE reflects systolic longitudinal LV function as a useful tool in the assessment of the LV function in a variety of situations that may evolve with risks for fetal heart failure, such as maternal DM. However, limited studies on the reference values of fetal MAPSE in low-risk population have been published until now [20,23]. Recently, reference values were established for fetal MAPSE, TAPSE, and septum annular plane systolic excursion (SAPSE) using 2D and M-mode between 20 and 36.6 weeks of gestation in low-risk pregnant women. The mean of each parameter ranged as follows: SAPSE, 2.87–5.56 mm; MAPSE, 3.98–8.07 mm; and TAPSE, 2.34–4.21 mm. There was a significant positive correlation between these parameters and GA [30].
To the best of our knowledge, this was the first study that assessed MAPSE using STIC-M in fetuses of mothers with pregestational DM. Atiq et al. [31] performed a study of fetal MAPSE using the conventional 2D M-mode in mothers with gestational diabetes (64 cases and 64 controls). The values of MAPSE were lower in the diabetic group than in the normal group (4.8 mm in DM vs. 5.0 mm in controls). Our results of fetal MAPSE using STIC-M were similar to those of Atiq et al. [31], with mean MAPSE values of 4.8 in the pregestational DM group and 5.0 mm in the control group. The differences in fetal MAPSE between the two groups reflect the effect of DM on LV function. In this study, significant differences between fetal MAPSE, TAPSE, and CO were observed in mothers with pregestational DM and healthy mothers, reflecting changes in the cardiac function.
The limitations of this study were the small number of cases. Additionally, the comparison between DM and control groups based on GA using Z-score or standard deviation were not done. In addition, we did not compare the values according to each trimester of pregnancy as well as between well and poorly controlled DM. Furthermore, the postnatal outcomes were not available.
In conclusion, fetal MAPSE, TAPSE, and CO using STIC-M and VOCAL could be applied and may assist fetal echocardiographers in the antenatal surveillance of fetuses with high-risk conditions for fetal heart failure, such as mothers with pregestational DM.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Ethical approval
The study was approved by the Ethic Committee of UNIFESP (CAAE: 87111116.4.0000.5505).
Patient consent
All participants provided a consent form.
Funding information
None.