Prophylactic tranexamic acid to reduce blood loss and related morbidities during hysterectomy: a systematic review and meta-analysis of randomized controlled trials
Article information
Abstract
To perform a systematic review and meta-analysis of all randomized controlled trials (RCTs) that evaluated the efficacy and safety of prophylactic tranexamic acid (TXA) versus a control (placebo or no treatment) during hysterectomy for benign conditions. Six databases were screened from inception to January 23, 2022. Eligible studies were assessed for risk of bias. Outcomes were summarized as weighted mean differences and risk ratios with 95% confidence intervals in a random-effects model. Five studies, comprising six arms and 911 patients were included in the study. Two and three studies had an overall unclear and low risk of bias, respectively. Estimated intraoperative blood loss, requirement for postoperative blood transfusion, and requirement for intraoperative topical hemostatic agents were significantly reduced in a prophylactic TXA group when compared with a control group. Moreover, postoperative hemoglobin level was significantly higher in the prophylactic TXA group than in the control group. Conversely, the frequency of self-limiting nausea and vomiting was significantly higher in the prophylactic TXA group than in the control group. There were no significant differences between the groups in terms of surgery duration, hospital stay, and diarrhea rate. All the RCTs reported no incidence of major adverse events in either group, such as mortality, thromboembolic events, visual disturbances, or seizures. There was no publication bias for any outcome, and leave-one-out sensitivity analyses demonstrated stability of the findings. Among patients who underwent hysterectomy for benign conditions, prophylactic TXA appeared largely safe and correlated with substantial reductions in estimated intraoperative blood loss and related morbidities.
Introduction
Hysterectomy is a commonly performed gynecologic procedure worldwide, and it is performed for a wide variety of benign and malignant conditions [1]. As a major procedure, hysterectomy is not free of perioperative complications [2], even when the indication for hysterectomy is a benign underlying cause [3,4]. Specifically, intraoperative bleeding is the most frequently encountered complication of hysterectomy [5], and is disadvantageously linked to higher morbidity and mortality if left unmanaged [6]. Thus, mechanical and pharmacological approaches that focus on reducing blood loss and related morbidities during hysterectomy are crucial.
Tranexamic acid (TXA), a man-made derivative of the amino acid lysine, is a common antifibrinolytic drug. From a mechanistic point of view, TXA blocks fibrinolysis and degradation of related blood clots. As a result, TXA stabilizes the fibrin meshwork and promotes anti-hemorrhagic effects [7]. Clinically, TXA has been shown to exhibit beneficial hemostatic effects for a wide variety of medical and surgical indications. Collectively, the accumulating body of literature highlights that TXA administration is feasible, safe, and effective in decreasing blood loss, transfusion need, and hemorrhage-related death [7–9].
However, the role of prophylactic TXA during hysterectomy has not yet been established. Few randomized controlled trials (RCTs) have examined the role of prophylactic TXA during hysterectomy. The results of these RCTs are conflicting and largely limited by their small sample sizes [3,5,10–12]. To the best of our knowledge, no meta-analysis has been conducted to comprehensively condense the literature on this subject. Such research is important for systematically gathering evidence and generating solid conclusions that can rationally inform clinical practice.
Hence, this study aimed to establish a meta-analysis of all RCTs that evaluated the clinical usefulness and safety of prophylactic TXA during hysterectomy.
Methods
1. Study protocol and registration
We carried out this investigation in line with the guidelines emphasized in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [13] in addition to the Cochrane Handbook for Systematic Reviews of Interventions [14]. We did not retrospectively register this investigation in the International Prospective Register of Systematic Reviews. Lastly, we did not seek ethical approval, as our investigation did not directly involve human or animal subjects, but rather utilized only published literature.
2. Eligibility criteria
We considered all studies that met the following evidence-based criteria: (i) patients: individuals undergoing hysterectomy; (ii) intervention: prophylactic TXA; (iii) comparator: any control treatment inclusive of placebo or no treatment; (iv) outcomes: reporting of any of our predetermined efficacy and safety endpoints; and (v) study design: RCTs that were published in peer-reviewed journals or found in selected grey literature that provided full-length reports, namely preprints and conference proceedings. We excluded all studies that met the following criteria: non-randomized studies, nonoriginal studies (for example, editorials, conference abstracts, and narrative review articles), and studies with procedures other than primary hysterectomy (for example, myomectomy).
3. Information sources, search strategy, and study selection process
Six databases were screened. We regarded five databases as primary information sources: Cochrane Central Register of Controlled Trials, PubMed, Embase, Scopus, and Web of Science. The sixth database (Google Scholar) was regarded as a secondary information source.
We utilized the following extensive query search in all the databases: (tranexamic acid OR transamine OR lysteda OR cyklokapron) and (hysterectomy) and (blood OR bleeding OR hemorrhage OR hemorrhage) and (randomized OR randomized OR randomly). We did not use any filters during the database search, such as the language, date of publication, or country of research. For primary information sources, we considered all citations retrieved from inception to January 23, 2022. Contrastingly, for the secondary information source, we considered only the first 200 citations retrieved from inception to January 23, 2022. Two coauthors independently performed the database search, and discrepancies were resolved by mutual agreement. Supplementary Table 1 shows the exact query search strategy used in all the databases.
Eligible studies were selected in three phases. First, duplicate citations were omitted. Second, we examined the titles and abstracts of the citations and discarded irrelevant citations. Third, we read the full text of the citations and made a verdict on the final inclusion in the systematic review and meta-analysis. To decrease the likelihood of missing relevant citations, we manually examined the reference lists of all eligible RCTs and recent narrative and systematic reviews. Two coauthors independently performed the database search, and discrepancies were resolved by mutual agreement.
4. Data items and collection process
We extracted information about the baseline characteristics of the included studies, namely author name, publication date, region of publication, study arms, sample sizes of participants, selection of patient demographics (i.e., age, body mass index, and indications of hysterectomy), type of hysterectomy, and details of prophylactic TXA intervention. Moreover, we extracted information about efficacy endpoints, namely, the mean estimated intraoperative blood loss (mL), mean postoperative hemoglobin level (g/dL), mean duration of surgery (minutes), mean hospital stay (days), rate of patients who required intraoperative hemostatic agents (%), and rate of patients who required postoperative blood transfusion (%). Additionally, we extracted information on safety endpoints, namely nausea and vomiting, diarrhea, abdominal pain, and major adverse events (e.g., mortality, thromboembolic events, seizures, and visual disturbances).
Relevant information was extracted using a pre-specified template. Moreover, we contacted the corresponding authors of some studies to obtain raw data and clarify some outcomes. Three groups (two co-authors per group) independently extracted the information, and discrepancies were settled by mutual agreement among the co-authors of each group.
5. Risk of bias assessment
We assessed the quality of the included studies in accordance with the Cochrane risk-of-bias assessment tool for RCTs [15]. This tool evaluates several sources of bias, such as selection, performance, detection, reporting, and attrition biases. We scored each source of bias as unclear, low, or high risk. Two authors independently assessed the risk of bias, and discrepancies were settled through mutual agreement.
6. Data synthesis and statistical analysis
We summarized continuous and dichotomous data as weighted mean difference (WMD) and risk ratio (RR) with a 95% confidence interval (CI). The data were analyzed using the inverse variance and Mantel-Haenszel methods. We used the random-effects (DerSimonian and Laird) model, as the eligible RCTs estimated different yet related intervention effects [16]. We assessed between-study heterogeneity in accordance with a Cochran’s Q test P-value of <0.1 [17] and Higgin’s I2 >50% [18]. We used Review Manager software version 5.4.0 (Cochrane, London, UK) to generate graphical forest plots. Additionally, we performed leave-one-out sensitivity analyses to gauge the robustness of the summary results by sequentially eliminating one RCT at a time and recalculating the summary effect sizes (WMD or RR) of the remaining RCTs. Moreover, we evaluated publication bias through the qualitative interpretation of funnel plots for asymmetry and quantified the results using Egger’s regression [19] and Begg’s rank [20] tests. We used STATA software version 17.0 (StataCorp LLC, College Station, TX, USA) to perform leave-one-out sensitivity analyses and examine publication bias. For all purposes, a two-tailed P-value of <0.05 was considered statistically significant. Two studies [5,10] reported a median, range (minimum-maximum), or interquartile range values, and we computed the essential mean and standard deviation values as described previously by Wan et al. [21]. One study [10] had two arms (intravenous and topical administration of prophylactic TXA), and we regarded each arm as a stand-alone RCT during meta-analysis, consistent with the published literature [22,23]. We labelled the TXA intravenous arm RCT as Sallam and shady. [10] (2019) IV, whereas the TXA topical arm RCT as Sallam and shady. [10] (2019) T.
Results
1. Summary of literature search and baseline characteristics of the included studies
Fig. 1 displays the Preferred Reporting Items for Systematic Reviews and Meta-Analyses flowchart. Overall, 555 citations were retrieved from the databases, of which 227 citations were excluded due to duplication. Of the remaining 328 citations, 319 were excluded after reading titles and abstracts. The remaining nine citations were subjected to fulltext reading, of which four were excluded for the following reasons: conference abstract (n=1) [24], procedures other than hysterectomy (n=1) [25], and RCTs with in-progress recruiting stage without published results on clinicaltrials.gov (n=2, NCT04760301 and NCT02911831). Finally, five studies, comprising six arms and 911 patients (TXA, 476 patients; control, 435 patients) were included in this systematic review and meta-analysis [3,5,10–12]. The studies were conducted in India (n=3) [3,11,12], Egypt (n=1) [10], and the USA (n=1) [5]. The indications for hysterectomy were benign conditions, including myoma, menorrhagia, metrorrhagia, chronic pelvic pain, dysfunctional uterine bleeding, endometrial hyperplasia, and uterovaginal prolapse [3,5,10–12]. The dose of intravenous TXA ranged from 10 mg/kg to 1 g maximum and was administered in five arms [3,5,11,12]. The dose of topical TXA was 2 g (intra-abdominal injection) and was administered in one arm [10]. Table 1 summarizes the baseline characteristics of the included studies.

The Preferred Reporting Items for Systematic Reviews and Meta-Analyses flowchart for literature search. RCT, randomized controlled trial.
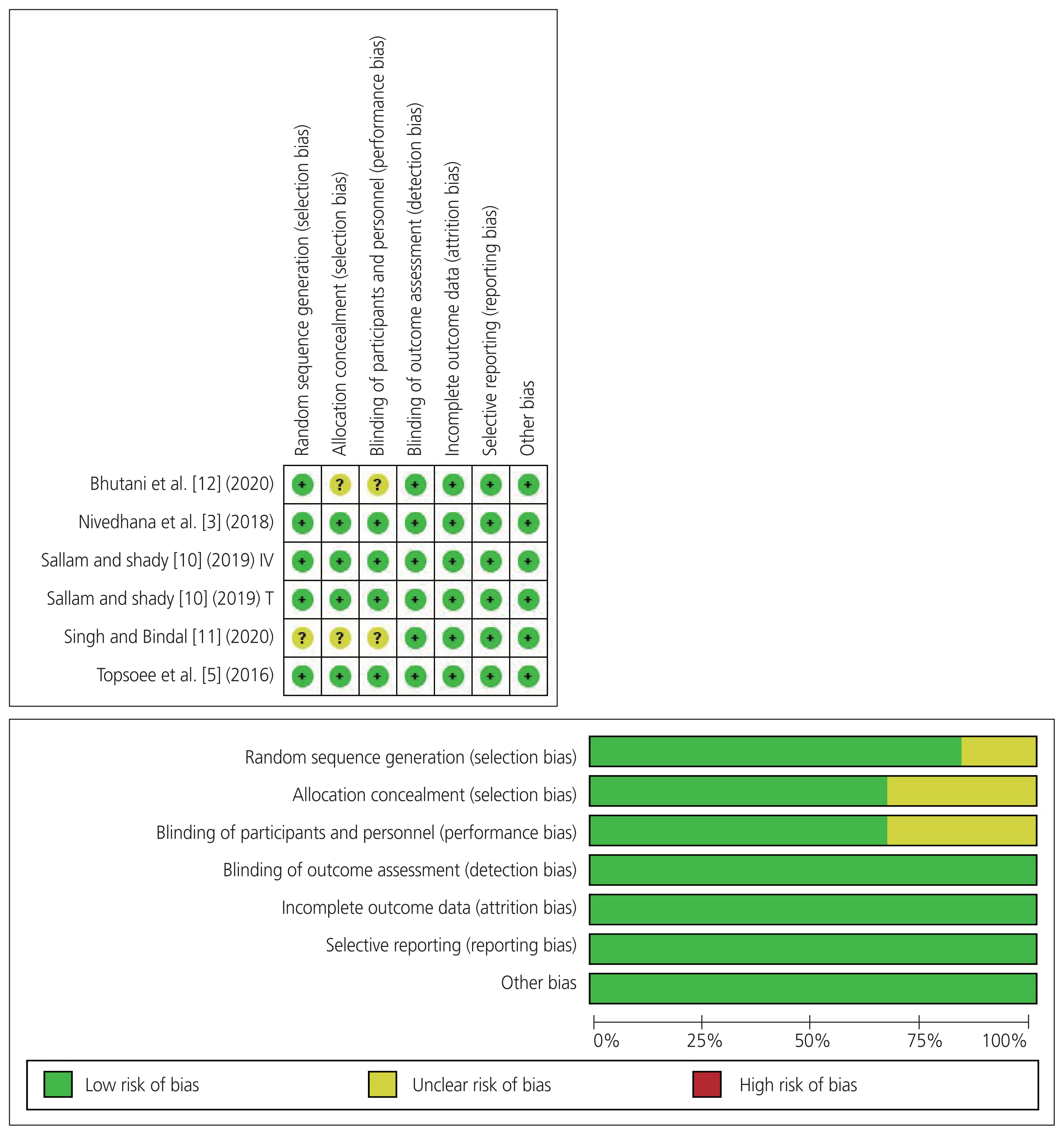
2. Summary of risk of bias of the included studies
One study [12] did not provide adequate information regarding allocation concealment and blinding of participants and personnel, and we graded the corresponding domains as unclear risk. Moreover, one study [11] did not provide adequate information about random sequence generation, allocation concealment, and blinding of participants and personnel, and we graded the corresponding domains as unclear risk. All remaining RCTs had a low risk of bias in all domains [3,5,10]. Overall, the included studies had a low risk of bias. Fig. 2 displays the risk of bias summary and a graph of the included studies.
3. Meta-analysis of efficacy endpoints
The pooled results revealed that the mean estimated intraoperative blood loss (n=6 RCTs; WMD=−143.15 mL; 95% CI, −190.21 to −96.09; P<0.001), requirement for postoperative blood transfusion (n=4 RCTs; RR=0.31; 95% CI, 0.16 to 0.58; P<0.001), and requirement for intraoperative topical hemostatic agents (n=2 RCTs; RR=0.41; 95% CI, 0.31 to 0.54; P<0.001) were significantly reduced in the prophylactic TXA group compared with the control group. The pooled results were homogeneous in terms of the requirement for intraoperative topical hemostatic agents (I2=0% and P=0.67), heterogeneous for mean estimated intraoperative blood loss (I2=82% and P<0.001), and requirement for postoperative blood transfusion (I2=89%, P<0.001). Moreover, the postoperative hemoglobin level was significantly higher in the prophylactic TXA group than in the control group (n=5 RCTs; WMD=0.61 g/dL; 95% CI, 0.27 to 0.95; P<0.001). The pooled results were homogeneous (I2=0%, P=0.94) (Fig. 3).
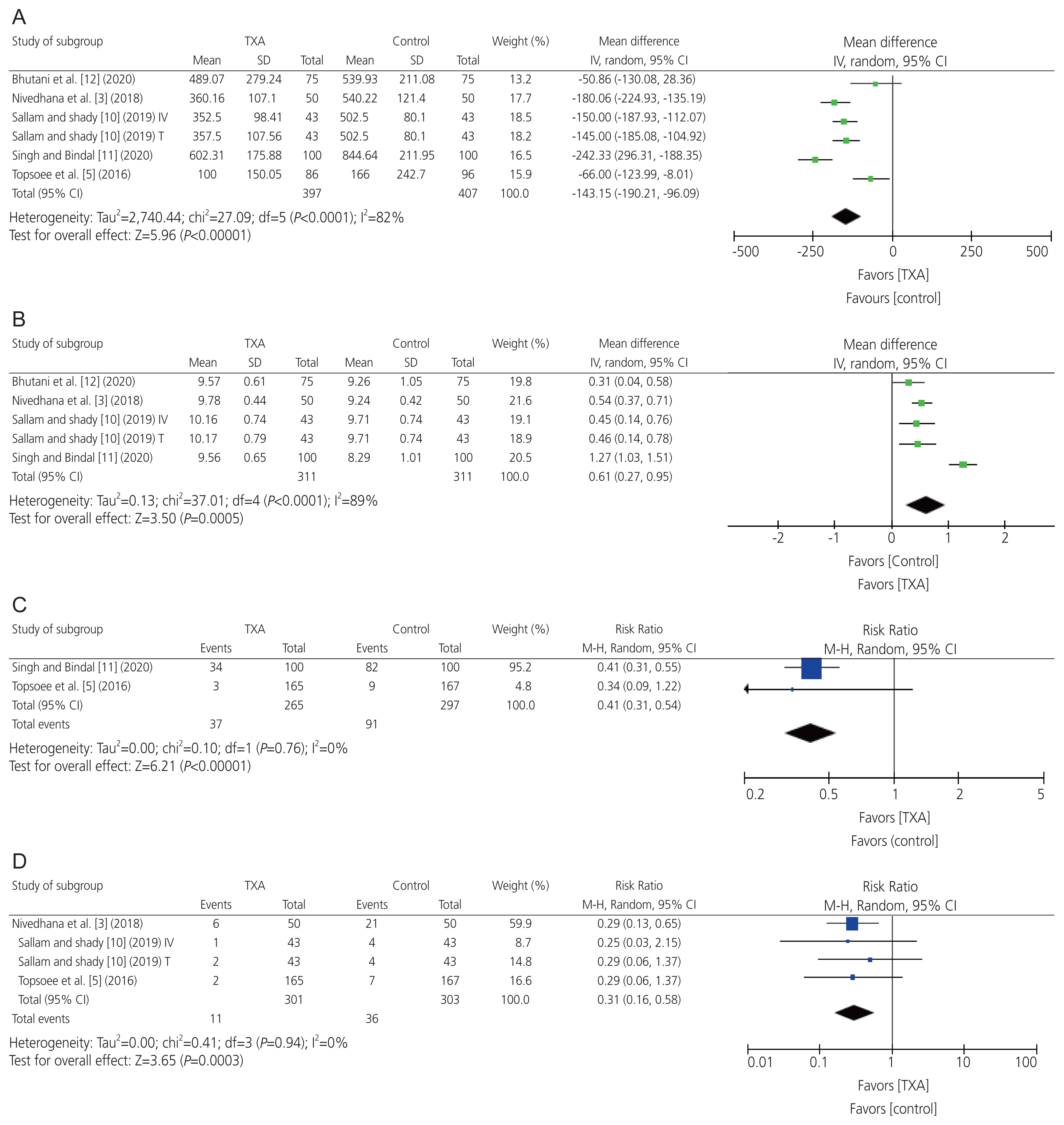
Meta-analysis of the efficacy endpoints: (A) mean estimated intraoperative blood loss, (B) mean postoperative hemoglobin level, (C) requirement rate for intraoperative hemostatic agents, and (D) requirement rate for postoperative blood transfusion. TXA, tranexamic acid; SD, standard deviation; CI, confidence interval; M–H, mantel-haenszel.
However, there was no significant difference between the groups regarding the duration of surgery (n=4 RCTs; WMD=−6.50 minutes; 95% CI, −17.55 to 4.56; P=0.25) and length of hospital stay (n=4 RCTs; WMD=−0.07 days; 95% CI, −0.33 to 0.19; P=0.58). The pooled results were heterogeneous (I2=95% and P<0.001) and homogeneous (I2=51%, P=0.1) (Fig. 4).
4. Meta-analysis of safety endpoints
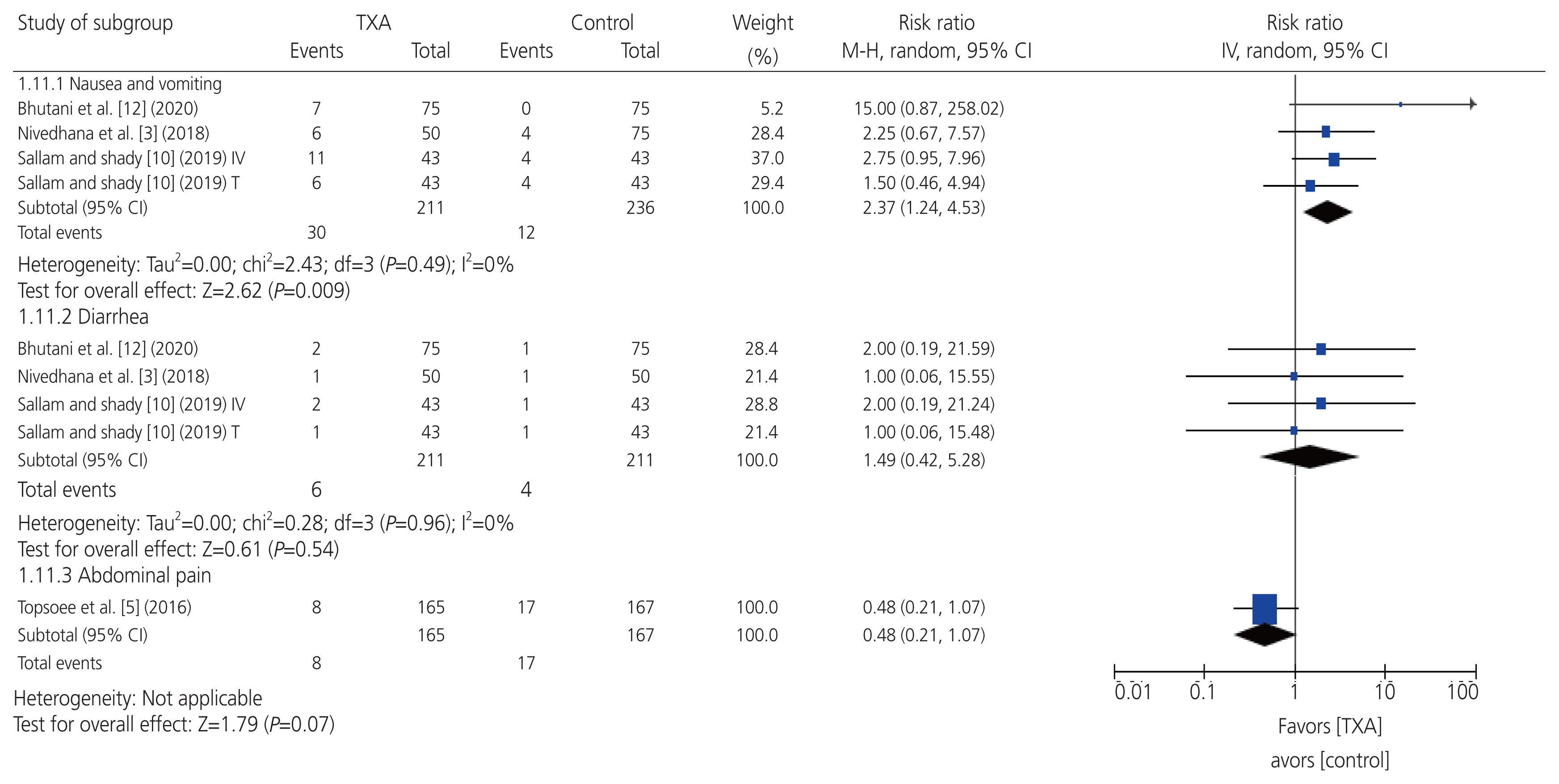
The pooled results revealed that the frequency of nausea and vomiting was significantly higher in the prophylactic TXA group than that in the control group (n=4 RCTs; RR=2.37; 95% CI, 1.24 to 4.53; P=0.009). Nevertheless, there was no significant difference between the groups in terms of the frequency of diarrhea (n=4 RCTs; RR=1.49; 95% CI, 0.42 to 5.28; P=0.54) or abdominal pain (n=1 RCT; RR=0.48; 95% CI, 0.21 to 1.07; P=0.07). All the pooled results were homogeneous (I2=0% and P=0.49, I2=89% and P=0.94, respectively) (Fig. 5). All the RCTs reported no incidence of any major adverse events in either group, such as mortality, thromboembolic events, visual disturbances, or seizures [3,5,10–12].
5. Leave-one-out sensitivity analysis
For the endpoint requirement for intraoperative hemostatic agents (n=2 RCTs; pooled RR=0.41; 95% CI, 0.31 to 0.54; P<0.001), omission of the Topsoee et al. [5] influenced the summary effect size (RR) to be statistically insignificant between both groups (RR=0.34; 95% CI, 0.09 to 1.22; P=0.098). Nevertheless, the results revealed stability and robustness for all remaining efficacy and safety endpoints, suggesting that individual studies did not have a significant impact on the summary effect sizes (Supplementary Figs. 1, 2).
6. Publication bias
For all efficacy and safety endpoints, the results revealed no significant asymmetry of funnel plots, and the quantitative P-values for the Egger’s regression test, and Begg’s rank test showed no statistical significance for publication bias (Supplementary Figs. 3, 4).
DISCUSSION
1. Summary of findings
This systematic review and meta-analysis aimed to summarize the clinical utility and safety of prophylactic TXA in patients undergoing hysterectomy. We included five studies, comprising six arms and 911 patients (TXA, 476 patients; control, 435 patients). The included studies had an overall low risk of bias. The pooled results showed that prophylactic TXA was associated with significant reductions in the mean estimated intraoperative blood loss, requirement for postoperative blood transfusion, and requirement for intraoperative topical hemostatic agents compared with the control group. Moreover, the pooled results showed that prophylactic TXA correlated with a significant increase in postoperative hemoglobin levels compared with the control group. Nevertheless, no significant difference was noted between the groups in terms of the duration of surgery and length of hospital stays. Regarding adverse events, the rates of self-limiting nausea and vomiting were significantly higher in the prophylactic TXA group than in the control group. Otherwise, prophylactic TXA appeared largely safe and did not culminate in major adverse events. Leave-one-out sensitivity analyses revealed stability and robustness for all the endpoints, except for the requirement rate for intraoperative hemostatic agents. There was no publication bias for any of the efficacy or safety end points.
2. Interpretation of findings and clinical implications
The mean estimated intraoperative blood loss was the major endpoint of efficacy during hysterectomy. Precise quantification of intraoperative blood loss is technically challenging. All included RCTs estimated intraoperative blood loss using the gravimetric method, which is less accurate, but is commonly used and regarded as the most practical method. Although the colorimetric (photometric) method is the most accurate method, its wide utilization is limited by complexity and cost issues [26].
Consistent with the present meta-analysis, uterine fibroids and menorrhagia are two of the most common indications for hysterectomy [27]. Accumulating evidence suggests that uterine fibroids are intrinsically characterized by richer vascularity in contrast to normal myometrium [28,29]. Moreover, menorrhagia has been shown to increase fibrinolytic activity [30,31]. These facts may be related to the relatively high frequency of bleeding-related complications encountered during hysterectomy. It is critical to control blood loss during hysterectomy. This is because substantial intraoperative blood loss may obfuscate the surgical field, increase the probability of sustaining iatrogenic injury, and culminate in unfavorable postoperative outcomes (e.g., infection, hindered wound healing, anemia, hemodynamic shock, and blood transfusion) [6]. Overall, patients undergoing hysterectomy may benefit from the administration of prophylactic blood-conserving pharmacological agents, such as TXA.
Several mechanical approaches, such as electrocautery, pressure application, and blood vessel ligation have been used to control intraoperative blood loss during hysterectomy. Nevertheless, topical and/or systematic blood-conserving pharmacological methods may be required to enhance hemostasis [32,33]. Generally, the three most frequently employed antifibrinolytic drugs during major surgery are aprotinin, ɛ-aminocaproic acid, and TXA [34]. Some of the major disadvantages of aprotinin include higher frequencies of renal injury and all-cause death than those of other antifibrinolytic drugs. Contrastingly, ɛ-aminocaproic acid is at least 6–10 times less potent than TXA [9,35]. Moreover, TXA advantageously does not affect blood coagulation indices [9], although one of its drawbacks is the requirement to adjust the dose among patients with renal insufficiency [35].
Superior evidence from several meta-analyses of RCTs has validated the clinical usefulness of prophylactic TXA in reducing perioperative bleeding, transfusion need, hospital stay, and mortality rate. These meta-analyses encompassed an extensive diversity of various operative procedures, including postpartum hemorrhage following Cesarean delivery [36], hepatic surgery [37], tonsillectomy [38], orthopaedic trauma surgery [39], endoscopic sinus surgery [40], and total-knee arthroplasty [41]. Our meta-analysis expands the clinical utility of prophylactic TXA in hysterectomy patients. Our data showed that prophylactic TXA reduced intraoperative blood loss, which was statistically significant and clinically meaningful. Amassing body of literature highlights that among women undergoing hysterectomy and myomectomy, the rate of preoperative anemia is high and represents an independent risk factor for 30-day postoperative serious aftermaths [42,43]. These hysterectomy and myomectomy procedures may culminate in significant perioperative blood loss, further aggravating the magnitude of coexisting anemia. For gynecologic patients with concomitant cardiovascular disease or severe anemia, minimal blood loss, for example, as low as 200 mL, may be lethal and warrant immediate rescue intervention. Additionally, this meta-analysis revealed that prophylactic TXA reduced the need for blood transfusion. This is very important, particularly in low-resource settings, where blood bank facilities are not readily available. Additionally, blood products are intrinsically regarded as scarce and are not free of potential transfusion-related hazards [44]. The overall conclusions of our meta-analysis are in line with the findings of a retrospective study by Jain and Shikha [45].
The mechanism of action of TXA is anti-fibrinolytic, which hypothetically implies that TXA is likely to promote blood clotting and increase mortality. However, high-quality evidence from a meta-analysis of 216 RCTs, comprising 125,550 patients corroborated that intravenous TXA, regardless of dosing, was not correlated with an amplified hazard of any thromboembolic event [46]. Rather, intravenous TXA was linked to a significant decrease in overall mortality and bleeding-specific mortality compared to the control (placebo/no treatment) intervention [46]. Overall, TXA is largely well-tolerated, with self-limiting nausea and vomiting being the most frequent side effects [9].
Sallam and Shady [10] reported that prophylactic topical and intravenous TXA were safe and effective in reducing perioperative blood loss and related morbidities in patients undergoing hysterectomy. A similar conclusion was obtained in a large meta-analysis of 18 RCTs involving patients undergoing various orthopedic surgeries [47]. Moreover, Teoh et al. [48] conducted an enormous meta-analysis of 71 RCTs, comprising 7,539 surgical patients. They reported that prophylactic topical TXA was safe and resulted in various clinical benefits (e.g., reduced intraoperative blood loss, total blood loss, hospital stay, and blood transfusion) compared with prophylactic topical placebo. Topical application of TXA has several advantages, such as feasibility, ease of administration, cost-effectiveness, avoidance of the first-pass effect of metabolism, and high local concentration of drugs with low potential for systemic toxicity [10].
3. Strengths and limitations
Our study has several strength points that ought to be emphasized. This is the first meta-analysis to comprehensively characterize the efficacy and safety of prophylactic TXA during hysterectomy. We considered only RCTs in our investigation to warrant the synthesis of data from high-quality studies. We screened five primary databases and searched Google Scholar (6th database) to select gray literatures to pool evidences from as many relevant RCTs that met our inclusion criteria. We performed a meta-analysis of several efficacy and safety endpoints to better inform clinical practice. We performed leave-one-out sensitivity analyses to explore the robustness of our pooled results. We also explored the publication bias using qualitative and quantitative methods.
Nevertheless, our study had several limitations that should be acknowledged. The small number of included RCTs and their corresponding small sample sizes represent a major drawback. Further drawbacks include a significant level of heterogeneity for some of the reported outcomes. This might be ascribed to reasons concerning methodological heterogeneity (e.g., variations in the surgical technique and dose of TXA) and clinical heterogeneity (e.g., comorbid characteristics of patients). Moreover, the included RCTs failed to report other important endpoints, such as estimated postoperative blood loss and long-term adverse events. Lastly, the data on publication bias should be interpreted with caution, owing to the reduced power of statistical tests to detect publication bias when the number of included studies is small (<10 studies per outcome) [19].
4. Future research directions
To further consolidate the summary findings of this meta-analysis, future research should include more high-quality and large-sized RCTs, comparing TXA with normal saline (placebo) among patients undergoing hysterectomy. Two RCTs are currently in progress (ClinicalTrials.gov, NCT04760301 and NCT02911831). A recent meta-analysis by Hafidh et al. [49] showed that prophylactic vasopressin administration reduced intraoperative blood loss compared with normal saline. Hence, future research should explore the clinical utility of intraoperative blood control during hysterectomy in a head-to-head comparison between TXA and other active competitors (e.g., vasopressin). Moreover, a recent meta-analysis by Sun et al. [50] showed that TXA administered in a combination fashion (intravenously and topically) versus either alone (intravenous or topical) resulted in higher efficacy in controlling bleeding without increasing the risk of thromboembolic events among patients undergoing total knee and hip arthroplasty. Hence, further research should investigate the clinical impact of different routes of TXA administration (intravenous versus topical versus combination) among patients undergoing hysterectomy. Bonus research may determine which cohorts of hysterectomy patients (stratified based on indications for hysterectomy and types of hysterectomy) are more likely to gain maximum benefit from prophylactic administration of TXA.
Conclusion
Among patients undergoing hysterectomy for benign conditions, prophylactic TXA correlated with substantial reductions in the mean estimated intraoperative blood loss and related morbidities. Moreover, prophylactic TXA appeared to be largely safe and free of any major adverse events compared to the control group. Nonetheless, additional RCTs are needed to corroborate the findings of this systematic review and meta-analysis.
Notes
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
No ethical approval was required because this review analyzed published data.
Patient consent
None.
Funding information
None.
Supplementary material
Supplementary Table 1 and Figs. 1–4 associated with this article can be found online at https://doi.org/10.5468/ogs.22115.