A novel clinicopathological analysis of early stage ovarian Sertoli-Leydig cell tumors at a single institution
Article information
Abstract
Objective
To evaluate the clinical and pathologic characteristics of patients who were diagnosed with ovarian Sertoli-Leydig cell tumors (SLCTs) in a single institution.
Methods
The medical records of 11 patients who were pathologically diagnosed with SLCTs beginning in 1995 in a single institute was reviewed.
Results
The median patient age was 31 years (range, 16 to 70 years). Patient International Federation of Gynecology and Obstetrics stages were IA, IC, and IIB in 3 (27.3%), 6 (54.5%), and 2 (18.2%) patients, respectively. Six patients (54.5%) had grade 3 tumors, 3 patients (27.3%) had grade 2 tumors, and 1 patient (9.1%) had a grade 1 tumor. Four patients without children underwent fertility-sparing surgery, and 7 patients had full staging surgery, including a hysterectomy and bilateral salpingo-oophorectomy, with a laparoscopic approach used in 3. Eight patients underwent pelvic lymph node dissection, and 8 patients were administered adjuvant chemotherapy consisting of bleomycin, etoposide, and cisplatin in 6 cases, a modified bleomycin, etoposide, and cisplatin regimen in 1 case, and a combined paclitaxel and cisplatin regimen in 1 case. Two patients died of disease and were re-diagnosed with Sertoli form endometrioid carcinoma. The other patients remain alive without recurrence at the time of reporting.
Conclusion
Our findings suggest that regardless of tumor stage or grade, ovarian SLCT patients have a good prognosis. Close observation and unilateral salpingo-oophorectomy would be beneficial for women who still wish to have children, while hysterectomy and bilateral salpingo-oophorectomy with adjuvant chemotherapy would be the optimal treatment in other cases. Furthermore, meticulous pathologic diagnosis is needed to develop a precise treatment strategy.
Introduction
Ovarian sex cord-stromal tumors are a heterogeneous group of ovarian tumors that comprise approximately 5% to 8% of all ovarian malignancies [1]. Sertoli-Leydig cell tumors (SLCTs), also known as androblastomas, are a subset of ovarian sex cord-stromal tumors that constitute less than 0.2% of all ovarian tumors [23]. SLCTs are the most frequently observed low-grade malignancies, although poorly differentiated tumors may behave more aggressively [4].
The tumors typically produce androgens, and clinical virilization is noted in 70% to 85% of patients [5678]. Signs of virilization include oligomenorrhea followed by amenorrhea, breast atrophy, acne, hirsutism, clitoromegaly, deepening voice, and receding hairline. Though frequently characterized by the androgen production, only 30% of patients display virilization or defeminization on presentation [2]. In particular, classic endocrine symptoms are rarely observed in retiform type SLCTs or those with heterologous elements [9]. Instead, these tumors are highly variable in their proportions of Sertoli cells, Leydig cells, and/or fibroblastic cells, and patients most often present with abdominal pain or distention.
Investigation of a large series of SLCT cases in a uniform manner is challenging for a single institute because of the paucity of cases [10]. In addition, varied pathology and tumor differentiation states make proper diagnosis and development of optimal treatment difficult [11121314]. An extended case series could provide valuable diagnostic and prognostic information, leading to precise therapeutic strategies.
This study aimed to evaluate the clinical and pathologic characteristics of patients who were diagnosed with ovarian SLCTs in a single institution. Patient outcomes and prognoses were investigated.
Materials and methods
The present study was approved by institutional review board of Yonsei University. The medical records of 11 patients who were pathologically diagnosed with SLCTs of the ovary were reviewed. All patients were treated in a single institute from 1995 to 2014. The collected data included age, past medical history, initial symptoms and signs, initial imaging findings, type of surgery, incision type, stage, histological type, chemotherapy regimens, and prognosis. Patients were staged according to the International Federation of Gynecology and Obstetrics (FIGO) classification of ovarian tumors [15]. Experienced pathologists who specialized in gynecologic oncology examined all specimens.
Based on a pathological review, the cases were divided into 1 of the 4 SLCT categories. Grade 1 tumors were well differentiated and contained a significant component of Leydig cells as well as Sertoli cells. Grade 2 tumors exhibited intermediate differentiation, and tumors were composed of immature Sertoli cells are typically arranged in a diffuse pattern. Grade 3 tumors were poorly differentiated. Those tumors were largely composed of tissue resembling an undifferentiated gonad and may resemble sarcomas. Other tumors had an unclassified heterologous element, and they were tumors of intermediate or poor differentiation that contained cell types foreign to the developing gonad [4].
For all patients, surgery was the initial treatment. For women who opted to spare fertility, a unilateral salpingo-oophorectomy was performed. Total hysterectomy and bilateral salpingo-oophorectomy with omentectomy, appendectomy, pelvic lymph node dissection, and para-aortic lymph node sampling were performed as a staging operation in other cases. Patients were administered postoperative adjuvant chemotherapy after surgery and were followed-up periodically using clinical, serologic, and radiological evaluations.
Results
The median age of the patients was 31 years, ranging from 16 to 70 years. The initial chief complaints can be divided into two main groups: menstrual disorders and nonspecific symptoms caused by ovarian masses. The largest group of patients (54%, 6/11) presented with nonspecific symptoms, including abdominal pain, palpable mass, and abdominal distension. Two patients experienced amenorrhea. There was a single case of oligomenorrhea and dysfunctional uterine bleeding each. Initial imaging findings suggested a malignant epithelial ovarian tumor in 6 cases, a germ cell tumor in 2 cases, and fibroma in 2 cases (Table 1).
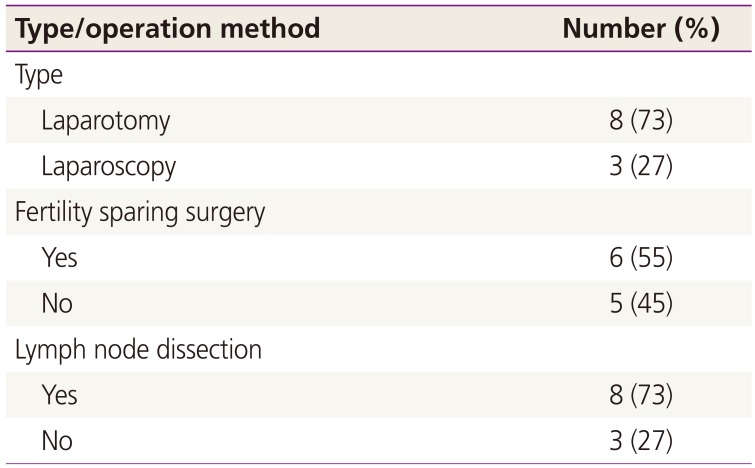
Grade 3 tumors were found in 6 patients (54.5%, 6/11), grade 2 tumors were found in 3 patients (27.3%, 3/11), and a grade 1 tumor was found in 1 patient (9.1%, 1/11). One patient had a tumor of unclassified grade (9.1%, 1/11), which was a pure stromal tumor composed of cells that resemble Leydig cells (Fig. 1). Eight patients were administered adjuvant chemotherapy after surgery, consisting of a bleomycin, etoposide, and cisplatin (BEP) regimen in 6 cases, a modified BEP regimen in 1 case, and combined paclitaxel and cisplatin in 1 case. Three patients who had IA/grade 2, IA/grade 3, or IC disease were not administered postoperative chemotherapy. Two of the 11 patients suffered disease recurrence and died of the disease 10 years after the operation. Of note, the tumors from these 2 patients were pathologically re-diagnosed as Sertoli-form endometrioid carcinoma (SEC). The other 9 patients remain alive without evidence of recurrence at the time of reporting (Table 2).
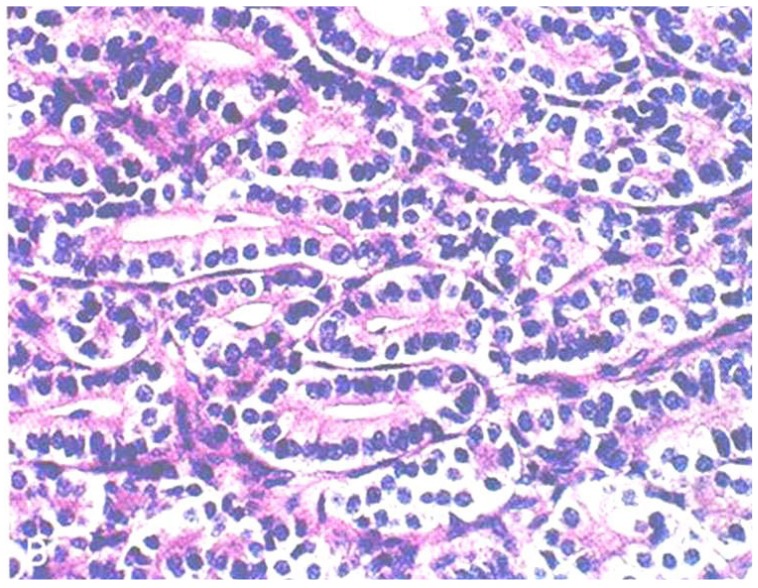
Pathology of Sertoli-Leydig cell tumor. There are solid tumors with round uniform cells vacuolated or pink granular cytoplasma and pigment (H&E, ×200).
Surgeries were performed as initial management for all 11 patients, and the 4 patients without children underwent fertility-sparing surgery. Seven patients had more extensive full staging surgery that included total abdominal or laparoscopic hysterectomy with bilateral salpingo-oophorectomy. Additionally, 8 of 11 patients underwent pelvic lymph node dissections. Three of the 11 patients underwent laparoscopic surgery, and 8 patients underwent laparotomies (Table 3). Tumors ranged from 2 to 20 cm in size and weighed 90 to 1,190 g. Most tumors had a smooth intact surface and were confined to the ovary (81.8%, 9/11), as seen in other series [16]. Three of the 11 patients (27.3%) had a tumor confined to one ovary (stage IA), 6 patients (54.5%) had tumors that ruptured during surgery (stage IC), and the other 2 patients (18.2%) presented with a tumor that had spread beyond the ovaries (stage IIb). No patients had bilateral disease (stage IB) (Table 1).
Discussion
SLCTs are a very rare type of sex cord-stromal tumor of ovary, accounting for less than 0.5% of all ovarian tumors [17], and little is known about them. SLCTs comprise diverse pathologic characteristics that can help pathologists and gynecologists make correct diagnoses, including primary and metastatic ovarian cancer [18]. In this study, patient age ranged from 17 to 70 years, and the average age was 31 years. SLCTs are more often encountered in young women. If a young woman has a pelvic mass and virilization, SLCT should be included in the differential diagnosis. Most of these tumors are unilateral and are diagnosed as stage I. Conservative surgery could be sufficient in young patients, and especially for those women who still wish to have children, a fertility sparing unilateral salpingo-oophorectomy and close observation could be beneficial. Hysterectomy and bilateral salpingo-oophorectomy, or full staging, with adjuvant chemotherapy would be the optimal treatment for women who do not need fertility preservation.
The stage and degree of tumor differentiation are the most important prognostic factors for these patients. For patients with poorly differentiated tumors, more aggressive management is needed because there is a high probability that these lesions will be malignant. An individualized treatment approach is necessary for patients with intermediately differentiated tumors. If patients have poor prognostic factors, adjuvant chemotherapy should be considered.
Owing to the rarity of this disease, there is still no consensus on the optimal management of SLCT or the most appropriate alternative therapy to treat stage I disease, which could involve chemotherapy, radiotherapy, and/or hormonal therapy. The BEP regimen is comparatively safe because it does not affect patient fertility status [19]. However, there has been no large-scale multicenter study to establish which chemotherapy regimen would be most effective, what the appropriate treatment would be for recurrence, or what the key prognostic factors are. A study by Brown et al. [20] in 2005 suggested an alternative regimen utilizing taxanes with or without a platinum agent, for the treatment of SLCTs.
To the best our knowledge, early stage SLCTs, such as well-differentiated stage IA tumors, are usually considered benign in South Korea, based on previous studies analyzing clinicopathological characteristics of the disease [2122]. According to the Korean standard disease classification, which is based on the World Health Organization classification, well-differentiated Sertoli-Leydig cell tumor is classified using disease code M8631/0. Code number 0 in the Korean classification specifies benign disease. Well-differentiated SLCT is considered benign, for which chemotherapy should not be administered. Among our cases, 1 case of well-differentiated SLCT was treated with chemotherapy due to accompanying stage IIB Meyer-Rokitansky-Kustner-Hauser syndrome. In our case series, 3 of 11 patients did not receive chemotherapy. Two of those patients were diagnosed with staged Ia disease, and the other was diagnosed with stage Ic disease. To date, all patients who were treated with surgery alone are alive. Chemotherapy use can be determined flexibly based on stage and patient performance status in SLCT treatment.
In our study, 8 of the 11 patients underwent platinum-based chemotherapy, and all except 2 are still alive at the time of reporting, including the 3 patients who did not undergo chemotherapy. Our findings suggest that, regardless of histologic grade or FIGO stage of ovarian SLCTs, the prognosis of these patients is generally excellent, even if no adjuvant chemotherapy is administered. Indeed, the 2 patients who succumbed to their disease were ultimately found not to have a SLCT. Sigismondi et al. [23] recommended in 2012 that platinum-based adjuvant chemotherapy be given to any patient with grade 2 to 3 disease, as prognoses for patients with disease stage >I and relapsed patients were poor.
First described by Young et al. in 1982 [24], SEC is a rare entity that is histologically similar to SLCT, which can result in even experienced pathologists misdiagnosing these tumors (Fig. 2). There are 2 different histologic points present in SEC contrary to SLCTs: (1) the parts with the typical pattern of endometrioid carcinoma and (2) the part of mucin at the apical borders of the tumor cells [25]. Even though they are similar in histology, there are important clinical differences between these two entities. SECs almost exclusively arise in postmenopausal women, with an average age of diagnosis between 60 and 70 years. In contrast, patients with SLCT tend to be younger, with an average age of diagnosis of 25 years, and may have hormonal signs, such as virilization [26]. Correspondingly, the 2 patients who were finally diagnosed with SEC in this study were relatively old.
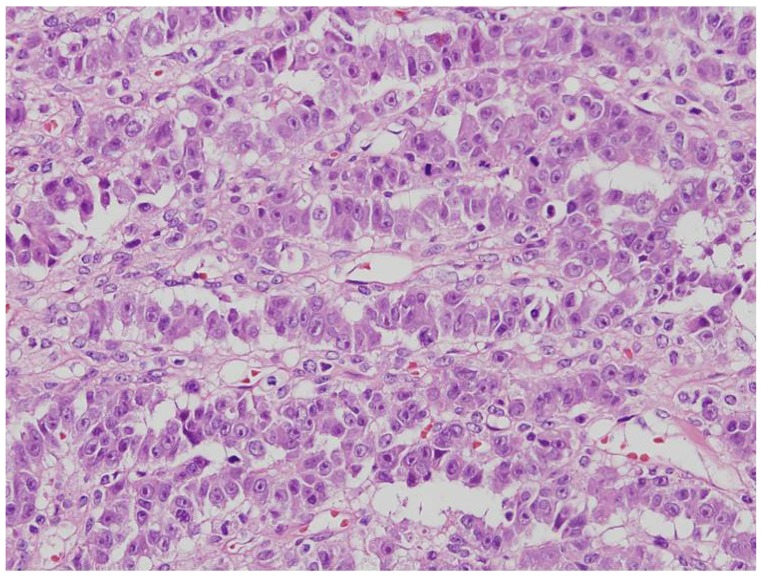
Sertoliform endometrioid carcinoma. The pathologic feature is similar to the Sertoli-Leydig cell tumors (H&E, ×200).
The majority of SECs described in past reports were initially misdiagnosed as SLCT [24]. Virilizing symptoms, though uncommon in endometrioid carcinoma, may be encountered in the Sertoli-form variant, making the diagnosis more difficult [25]. Taken together, this suggests that extensive sampling of the specimen and close investigation is necessary before making a diagnosis of SLCT, especially in postmenopausal patients. Recognition of this tumor is important, as it is a well-differentiated, low-grade malignancy that is associated with a good prognosis when confined to the ovary [2527]. On the other hand, SEC areas are usually classified as grade 3 based on the current FIGO grading system [27]. The two patients who expired in our study had poor prognoses.
There have also been a few case reports of successful laparoscopic removal of these tumors [28]. Laparoscopic surgery was performed in 3 patients in this study, and the patients all had equivalent outcomes to patients who underwent laparotomies. Laparoscopic surgery would be preferable in most cases, as it is minimally invasive, leaves less scaring, and is associated with a quicker recovery.
In the present study, we investigated clinical and pathologic characteristics of patients who were diagnosed as ovarian SLCTs in a single institution. Additionally, we evaluated patient outcomes and prognoses. Initial chief complaints related with virilization were present in only 4 cases, which was consistent with a previous report [2]. Patient prognoses were favorable in our study, even for patients did not receive adjuvant chemotherapy, and laparoscopy was a feasible approach to treat disease. A multi-centered study with more patients will be required to better understand SLCTs.
In conclusion, conservative surgery in a young patient who still wishes to have children is sufficient. Hysterectomy and bilateral salpingo-oophorectomy, or full staging, with adjuvant chemotherapy is the best treatment for patients who do not wish to spare fertility. However, if the patient, pathologically, has well-differentiated cell stage Ia disease, then chemotherapy is not necessary, as that is considered as benign disease.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.